More results...

In chemistry, understanding fundamental concepts such as the atomic number, and the mass number are essential. Before we move in to learn about those concepts, it is important to get a basic understanding of the fundamental particles (subatomic particles) of an atom.
Elements are made out of atoms. An atom can be defined as the smallest unit that an element can be divided. In other words, an atom is the smallest unit that resembles the chemical and physical properties of an element.
Atoms are made out of fundamental subatomic particles, electrons, protons, and neutrons. Those subatomic particles further can be divided into much smaller units. But we are not going to discuss them in this basic introduction.
When considering subatomic particles, neutrons, and protons are contained in the nucleus of an atom. Electrons occupy the region of space around the nucleus.
The mass of protons and neutrons are approximately similar, and the mass of an electron is negligible when compared to neutron and proton. That means nearly all the mass of an atom is concentrated in its nucleus.
The radius of a nucleus is about 10-15 m. When comparing this with the radius of an atom this is a very small fraction. So, the volume of the nucleus of an atom is a small fraction of the volume of an atom. That means the density of the nucleus is very high.
Eg: The atomic radius of a Hydrogen atom is about 3.7×10-11. This is nearly 104 times the radius.
Protons are positively charged, and neutrons are neutral. Electrons are negatively charged, and the magnitude of the charge is equal to the charge of protons.
| Subatomic particle | Charge – in Coulombs (C) | Relative charge (Charge number) | Rest mass (kg) | Relative mass |
| Proton | +1.602×10-19 | +1 | 1.673*10-27 | 1837 |
| Neutron | 0 | 0 | 1.675*10-37 | 1839 |
| Electron | -1.602×10-19 | -1 | 9.109*10-31 | 1 |
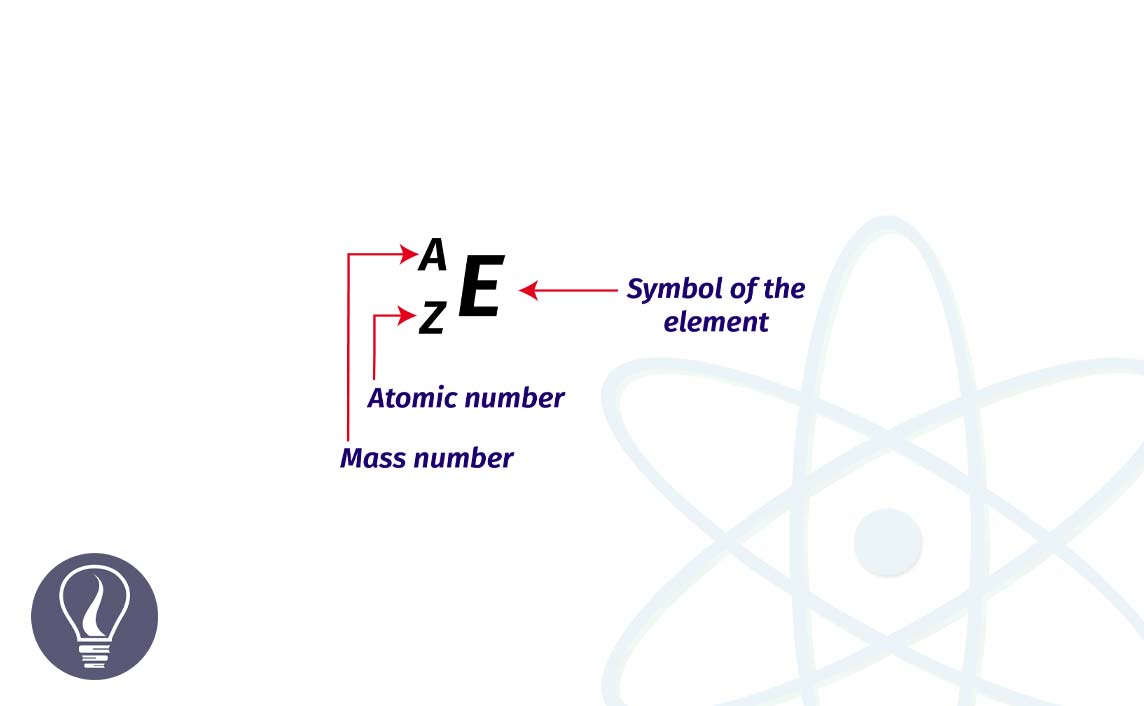
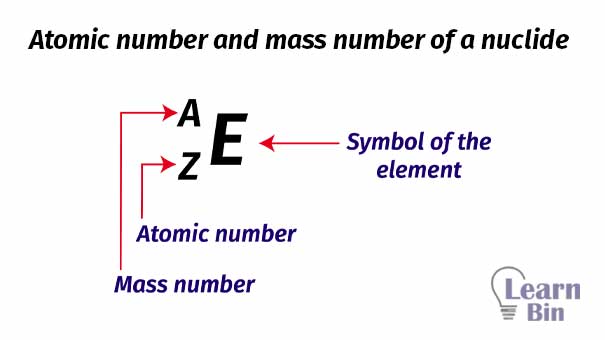
A Nuclide can be defined as a specific type of atom (or nucleus) that possesses a characteristic atomic number and a characteristic mass number. This is represented by the symbol ‘E’ in the shorthand method.
The atomic number of a nuclide (atom) is equal to the number of protons present in the nucleus of the atom. Because, in the ground state atom is electrically neutral, its atomic number is also equal to the number of electrons in the atom. The atomic number is represented by the symbol ‘Z’ in the shorthand method.
Atomic number = Z = Number of protons in nucleus = number of electrons (at ground state)
The mass number of the nuclide (atom) is equal to the total number of neutrons and protons in the nucleus. In the shorthand method, the mass number is represented by the symbol ‘A’.
Mass number = A = Number of protons in nucleus + number of neutrons in nucleus
Number of neutrons = Mass number – Atomic number = A - Z
Eg:
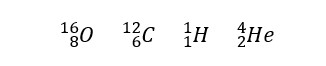
To learn about concepts of mass calculations of atoms (Atomic mass unit, relative atomic mass), you can visit the linked article.

Housecroft, C.E. and Sharpe, A.G. (2018) Inorganic Chemistry. Harlow: Pearson Education.
The cover image was created using an image by Dede from Pixabay