More results...


The Kjeldhal method is a quantitative analytical technique that is used to analyze the Nitrogen content of a sample. This method was developed by a Danish chemist, Johan Kjeldahl in 1883.
Using the Kjeldhal method it can be analyzed,
But without any modifications/ catalysts, other forms of inorganic nitrogen such as nitrates (NO3-), and nitrites (NO2-) cannot be analyzed in this method.
There are three steps involved in a chemical analysis carried out by the Kjeldhal method.
In the digestion step, all the Nitrogen present in the sample is converted into Ammonium sulfate. The sample is treated with conc. Sulfuric acid (H2SO4) in 360 - 410 0C.
This process is very slow and requires a lot of heat energy. Therefore, the sample needs to be added a catalyze. There are three types of catalyzes that are used in the digestion process in the Kjeldhal method.
I. Mercury based catalyzes
Mercury-based catalyzes have the highest rate of reaction. But when Hg-based catalyzes are used, all the Nitrogen present in the sample is not converted into Ammonium sulfate. Because some amount of Nitrogen undergoes side reactions and results in cyclic compounds that contain Nitrogen. To release nitrogen from these cyclic compounds, Sodium thiosulfate is added to the final mixture.
II. Selenium based catalyzes
Selenium-based catalyzes have a moderate reaction rate. But when selenium-based catalyzes are used, some amount of Nitrogen is converted to Nitric acid.
III. Copper-based catalyzes
The reaction rate is very low when the Copper-based catalyzes are used. Also, all the Nitrogen is not converted into Ammonium sulfate. Only a 98% recovery can be expected. Here, CuSO4 is formed. Therefore, a light blue color solution is obtained.
After the digestion is completed, a colorless solution is obtained. If there is color present, that indicates an incomplete digestion.
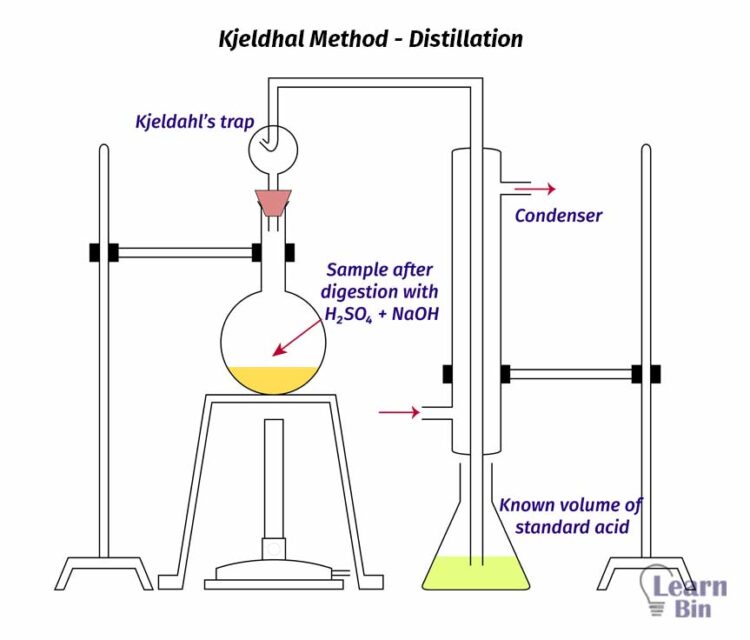
The solution of Ammonium sulfate that is obtained from the digestion process is transferred to a distillation unit. Before the distillation starts, 8M NaOH solution is added to the above solution. Then the solution is heated using steam. When heating Ammonium sulfate (NH4OH) is reacted with Sodium Hydroxide (NaOH) as follows.
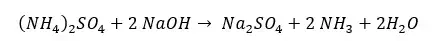
In this reaction, Ammonia gas (NH3) is released. This ammonia is trapped in 4% Boric acid.
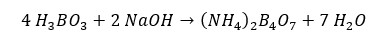
The collected solution from the distillation is then titrated with 0.2 M HCl solution in the presence of a Tashiro indicator.
Tashiro indicator is a mixture of two indicators. It is composed of 0.1% methylene blue and 0.03% methyl red dissolved in ethanol or methanol. At the endpoint, it gives a pinkish-red endpoint.
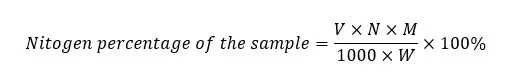
Where,
Therefore, the above equation can be arranged as follows.


The cover image was created using an image by Michal Jarmoluk from Pixabay
Figure 01: Contains an image by Roshan220195, licensed under CC BY-SA 3.0, via Wikimedia Commons