More results...


Oxides of sulfur are chemical compounds formed by the reaction of sulfur with oxygen. Sulfur forms two stable oxides which are sulfur dioxide (SO2) and sulfur trioxide (SO3). Both are at gaseous state at room temperature and these gases are significant both environmentally and industrially.
Sulfur dioxide is released from volcanic eruptions and the burning of fossil fuels, contributing to air pollution and acid rain. Sulfur trioxide is important in the production of sulfuric acid, which is a key industrial chemical.
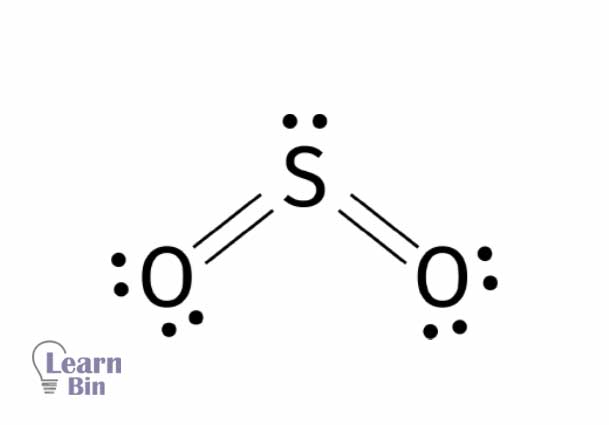
Sulfur dioxide is a colorless, toxic, and acidic gas with a pungent odor. The oxidation state of sulfur in sulfur dioxide is +4. Sulfur forms two double bonds with two oxygen atoms resulting in sulfur dioxide. Sulfur dioxide is an angular-shaped molecule with a lone pair of electrons on the sulfur atom. This makes the sulfur dioxide molecule a polar molecule.
Sulfur dioxide readily dissolves in water and results in sulfurous acid. (H2SO3). Sulfurous acid is a weak diprotic acid. That means in water it partially dissociates and results in two protons (H+ ions) to the water. Also, sulfur dioxide reacts with strong bases like Sodium Hydroxide (NaOH) to result in its respective sulfite and water. The following reactions explain the acidic properties of sulfur dioxide.
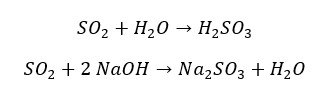
Sulfur dioxide shows both oxidizing and reducing properties.
The sulfur atom in sulfur dioxide possesses a +4 oxidation state. Sulfur dioxide shows reducing properties when it reacts with oxidizing agents. Here, the oxidation number of sulfur increases to +6. So, SO2 will be turned into Sulphate ion (SO42-). Thus, the SO2 will be oxidized while the other substance will be reduced.
SO2 can reduce MnO4– ion into Mn2+ ion in the presence of a dilute acid. SO2 gas oxidizes into sulfate ions. The oxidation number of sulfur changes +4 to +6 and the oxidation number of Mn Changes +7 to +2. MnO4- ion is purple in color and Mn2+ is colorless in an acidic medium. Here, the purple color KMnO4 solution will be turned colorless.
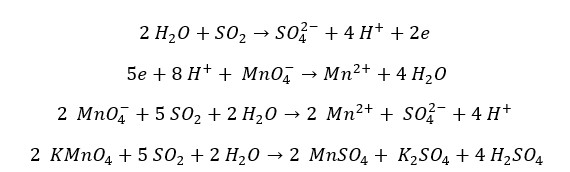
In an acidic medium SO2 reduces Cr2O72- ions into Cr3+ ions. SO2 gas oxidizes into sulfate ions. Here, the oxidation state of Cr changes from +6 to +3, and the oxidation number of sulfur changes from +4 to +6. In an acidic medium Cr2O72- ion is orange in color and the Cr3+ ion is green in color. Therefore, the orange color solution turns green in this reaction.
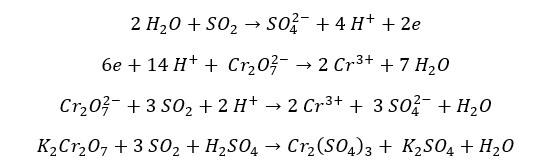
In the presence of dilute acid, SO2 gas reduces Fe3+ into Fe2+ and SO2 gas oxidizes into sulfate ion. The oxidation number of sulfur changes +4 to +6 and the oxidation number of Fe changes +3 to +2. In an acidic medium, Fe3+ is yellow-brown in color and Fe2+ is pale green in color. Therefore, the solution turns from yellow-brown to pale green color.
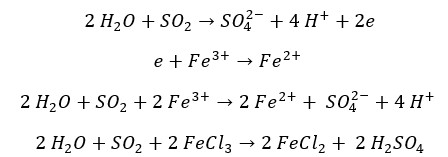
Hydrogen peroxide is a strong oxidizing agent. In the presence of dilute acid, SO2 gas reduces H2O2 into H2O and SO2 gas oxidizes into sulfate ion. The oxidation number of sulfur changes +4 to +6 and the oxidation number of Oxygen changes -1 to -2.
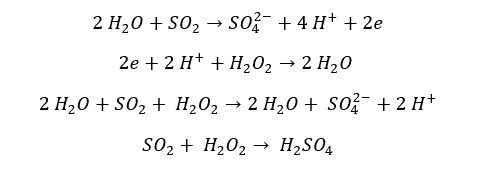
SO2 reduces halogens (Cl2, Br2, I2) into their respective anions. SO2 gas oxidizes into sulfate ions. The oxidation number of sulfur changes from +4 to +6 and the oxidation number of Cl, Br, or I change from 0 to -1. In the reaction with Br2, it can be observed that the red color of the Br2 liquid turns in to colorless.
Reaction with Cl2
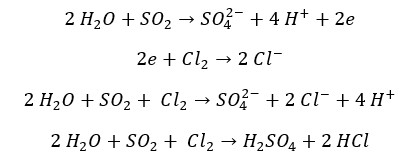
Reaction with Br2
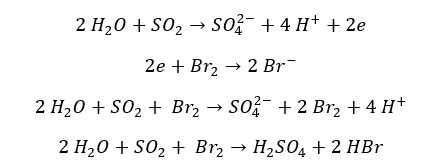
Reaction with I2
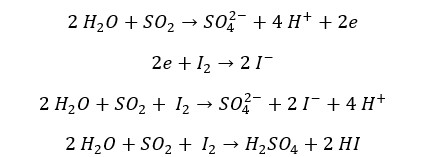
Wet flower petals are bleached in contact with SO2 gas. In the presence of water SO2 forms sulfurous acid. This sulfurous acid will further react with water to form sulfuric acid and atomic hydrogen. These atomic hydrogen are highly reactive and they can turn the pigments in the flower into their respective hydride. If the water dries up, it brings back its original color. The bleaching property of SO2 is also a reducing reaction of SO2.
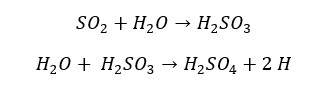
SO2 can act as an oxidizing agent when it is reacted with strong reducing agents like alkali metals, alkali earth metals, etc. Here, the oxidation state of sulfur changes from +4 to les oxidation state that could be +2 or 0.
H2S is a stronger reducing agent than SO2. Therefore, in the reaction between SO2 and H2S, SO2 Reduces into Sulfur (S) and H2S oxidizes into sulfur (S) also. The oxidation number of the sulfur atom in the SO2 changes +4 to 0 and the oxidation number of the sulfur atom in the H2S changes from -2 to 0. From this reaction, it forms milk of sulfur. Therefore, the solution will appear as milky white or pale yellow in color.
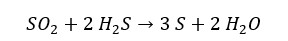
When Magnesium (Mg) is heated with SO2, Mg will oxidize into Mg2+ and SO2 will reduce into S. If there is excess of Mg, SO2 will be reduced to a Sulfide ion that has the oxidation state of -2.
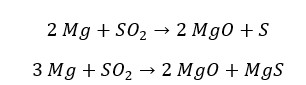
Industrially SO2 is prepared by burning sulfur in the excess of oxygen. In the laboratory, SO2 can be generated by adding a dilute acid to sulfites. Also, Hot Concentrated sulfuric acid will reduce into sulfur dioxide when it is reacted with some metals.
Burning of sulfur
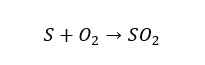
Reaction of sulfites and dilute acid
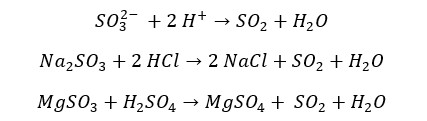
Reaction of hot concentrated sulfuric acid
A filter paper damped with acidic potassium dichromate (K2Cr2O7) turns green from an orange color in contact with sulfur dioxide gas.
A filter paper damped with acidic potassium permanganate (KMnO4) turns colorless from purple color in contact with sulfur dioxide gas.
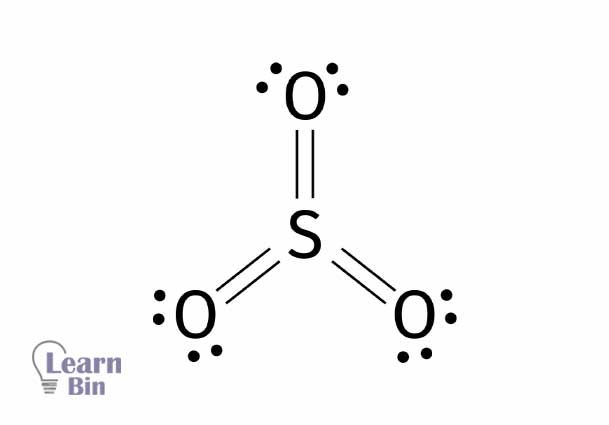
Sulfur trioxide (SO3) is a colorless and strongly acidic gas. The oxidation state of sulfur in sulfur trioxide is +6. Sulfur forms three double bonds with three oxygen atoms resulting in sulfur trioxide. Sulfur dioxide is a trigonal planner-shaped molecule with no lone pair electrons on the sulfur atom. This makes the sulfur dioxide molecule a non-polar molecule.
When SO3 is exposed to air it will react with the moisture in the atmosphere and form sulfuric gas. It appears as a mist. Unlike sulfur dioxide, sulfur trioxide reacts with water explosively resulting in sulfuric acid which is a strong diprotic acid. SO3 is mainly used in industries to produce sulfuric acid.
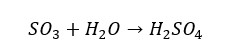
Sulfur trioxide can be prepared from the reaction with sulfur dioxide and oxygen gas.
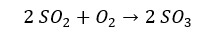

The cover image was created using an image by Andre Nadj from Pixabay