More results...

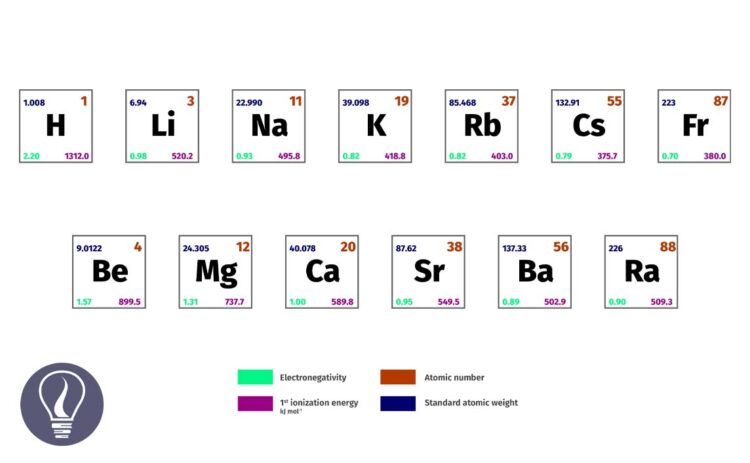
Group 2 elements, also known as alkali earth metals are comprised of s block of the periodic table. Except for Beryllium (Be), all the group 2 elements are metals. Beryllium is an amphoteric element that has both metallic and non-metallic properties.
When compared to group 1 elements, group 2 elements show less reactivity. The reactivity of group 2 elements is less than group 1 elements because the valance electrons of group 2 elements are tightly attracted by the nucleus of the atom. Because of this phenomenon, the effective nuclear charge is high.
Therefore, it needs higher energy to remove electrons from the valance shell in order to form ions. However, the reactivity increases down the group. All the group 2 elements remove two electrons from its valance shell and form +2 ions.
All the group 2 elements react with Oxygen and produce the respective oxide. When Beryllium and Magnesium are exposed to air, a thin layer of beryllium oxide and magnesium oxide form on their surfaces respectively.
When an oxide layer is formed on the surface, it acts as a barrier to oxygen, and the further reaction with oxygen stops. The oxide layer formed on the beryllium is thinner than the oxide layer formed on the magnesium.
Calcium (Ca), Strontium (Sr), and Barium (Ba) readily react with oxygen and form the respective oxide. The reactivity increases down the group.
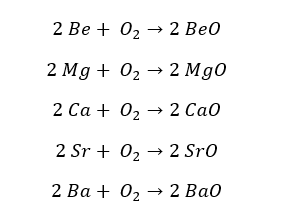
Barium forms its peroxide in the presence of excess Oxygen.
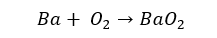
Group 2 elements don't react with nitrogen under room temperature at atmospheric pressure.
But at high temperatures, group II elements react with atmospheric Nitrogen gas. They form the respective nitrite. The reactivity increases down the group. The nitrides of group 2 elements are white color powder.
When going down the group reactivity of the elements increased, so the temperature required for the reaction reduced.
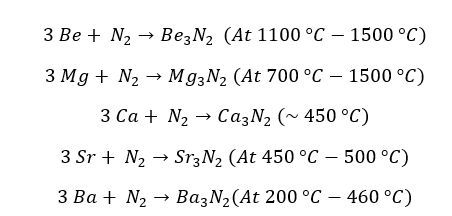
These nitrites can further react with water resulting in the respective hydroxide and ammonia gas.
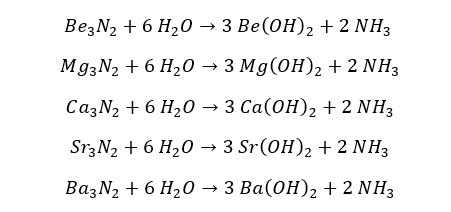
Except for Beryllium, all the group II elements react with Hydrogen gas (H2) and form the respective hydride.
Calcium (Ca), Strontium (Sr), and Barium (Ba) react with hydrogen gas at high temperatures. Magnesium (Mg) reacts with Hydrogen gas to form hydride at high temperatures and high pressures.
The oxidation state of the Hydrogen in these hydrides is -1.
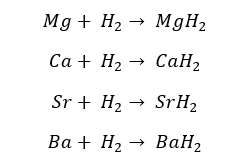
These hydrides of group II elements further react with water and form respective hydroxides of the element and hydrogen gas.
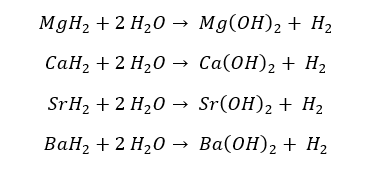
Group II elements react with Chlorine gas and produce the respective chlorides.
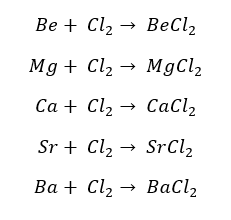
Except for Beryllium, all the group 2 elements react with water and form their respective hydroxides and hydrogen gas.
Beryllium does not react with liquid water but it reacts with steam at high temperatures (water vapor). Magnesium reacts with water, but at room temperature this reaction is negligible. Magnesium reacts with hot water resulting in magnesium hydroxide and hydrogen gas.
Calcium (Ca), Strontium (Sr), and Barium (Ba) readily react with water. The reactivity of this reaction increases down the group.
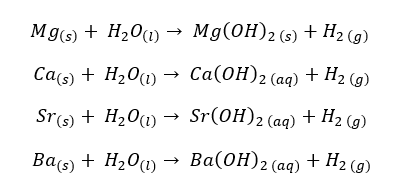
Beryllium reacts with steam at high temperatures (around 700 °C). Beryllium and Magnesium produce their oxides and hydrogen gas from the reaction with steam.
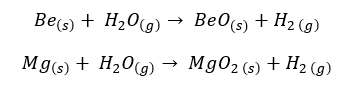
Group 2 elements readily react with the diluted acids and produce Hydrogen gas and the respective salts.
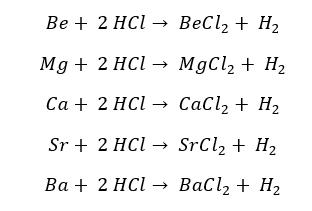
Group 2 elements react with concentrated acids as follows.
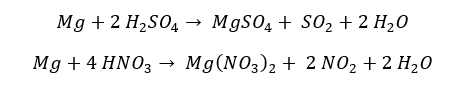

National Institutes of Health | PubChem - Periodic Table of Elements