More results...
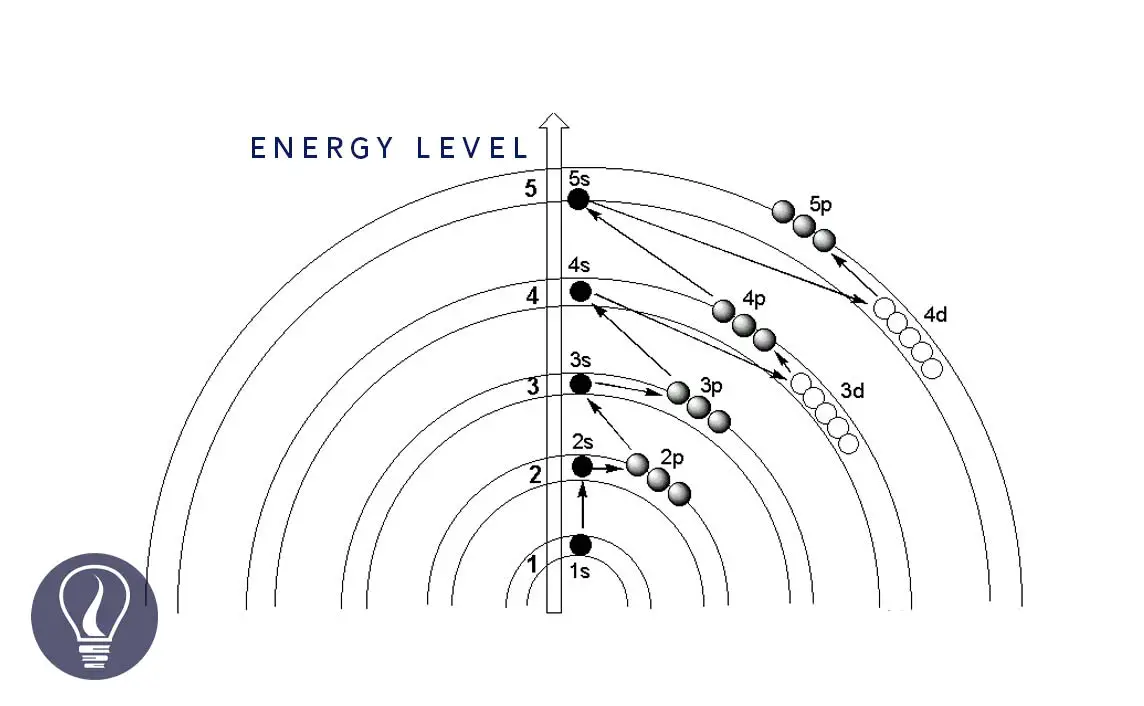

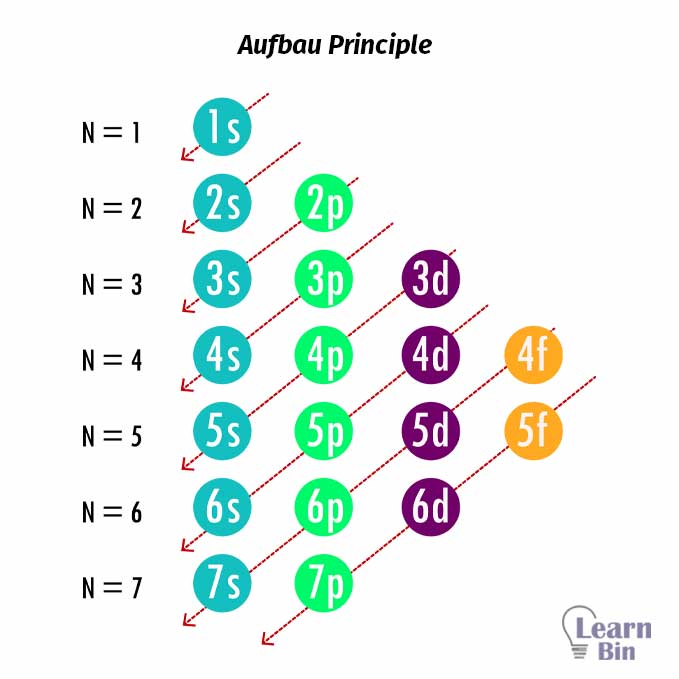
Electron configuration is a standard notation that describes the distribution of electrons that are present in the shells, subshells, and orbitals of an atom. The electron configuration is written according to the Aufbau principle.
The Aufbau principle states that the orbitals are filled with electrons from the lowest energy levels to higher energy levels. That means the lower energy levels are filled first. According to the Aufbau principle, the pattern that fills the orbitals can be represented in Figure 01.
But there are some instances that some elements deviate from the Aufbau principle.
| Elements | Atomic number | Electron configuration |
| H | 1 | 1s1 |
| He | 2 | 1s2 |
| Li | 3 | 1s2 2s1 |
| Be | 4 | 1s2 2s2 |
| B | 5 | 1s2 2s2 2p1 |
| C | 6 | 1s2 2s2 2p2 |
| N | 7 | 1s2 2s2 2p3 |
| O | 8 | 1s2 2s2 2p4 |
| F | 9 | 1s2 2s2 2p5 |
| Ne | 10 | 1s2 2s2 2p6 |
| Na | 11 | 1s2 2s2 2p6 3s1 |
| Mg | 12 | 1s2 2s2 2p6 3s2 |
| Al | 13 | 1s2 2s2 2p6 3s2 3p1 |
| Si | 14 | 1s2 2s2 2p6 3s2 3p2 |
| P | 15 | 1s2 2s2 2p6 3s2 3p3 |
| S | 16 | 1s2 2s2 2p6 3s2 3p4 |
| Cl | 17 | 1s2 2s2 2p6 3s2 3p5 |
| Ar | 18 | 1s2 2s2 2p6 3s2 3p6 |
| K | 19 | 1s2 2s2 2p6 3s2 3p6 4s1 |
| Ca | 20 | 1s2 2s2 2p6 3s2 3p6 4s2 |
| Sc | 21 | 1s2 2s2 2p6 3s2 3p6 4s2 3d1 |
| Ti | 22 | 1s2 2s2 2p6 3s2 3p6 4s2 3d2 |
| V | 23 | 1s2 2s2 2p6 3s2 3p6 4s2 3d3 |
| Cr | 24 | 1s2 2s2 2p6 3s2 3p6 4s1 3d5 |
| Mn | 25 | 1s2 2s2 2p6 3s2 3p6 4s2 3d5 |
| Fe | 26 | 1s2 2s2 2p6 3s2 3p6 4s2 3d6 |
| Co | 27 | 1s2 2s2 2p6 3s2 3p6 4s2 3d7 |
| Ni | 28 | 1s2 2s2 2p6 3s2 3p6 4s2 3d8 |
| Cu | 29 | 1s2 2s2 2p6 3s2 3p6 4s1 3d10 |
| Zn | 30 | 1s2 2s2 2p6 3s2 3p6 4s2 3d10 |
Cr and Cu deviate from the Aufbau principle. Because the d orbitals stabilize when they are completely or a half with electrons.
Atoms form positively charged ions (cations) by removing electrons. When atoms remove 1, 2, or 3 electrons respectively, they will form +1, +2, or +3 ions respectively. Thus, the electrons are removed starting from the highest energy orbital in the highest energy level to the lowest.
As an example, Sodium (Na) removes one electron and forms a +1 charged ion. This electron is removed from the 3s orbital which is the highest energy orbital that Sodium has.
But there are some atoms where the d orbitals are filled after the s orbital of the next energy level is filled. As an example, in copper atoms, the 3d orbital is filled after the 4s orbital. In such cases, electrons are removed from the 4s orbital first.
Atoms form negatively charged ions (anions) by gaining electrons. When atoms remove 1, 2, or 3 electrons respectively, they will form -1, -2, or -3 ions respectively. Electrons are filled to the last subshell of the valance shell.

The cover image was created using an image by Chris Evans, Dr. Roger Peters, Dr. Mike Thompson, Chris Gadsby, Ken Partridge, Roy Mylan, Yehoshua Sivan, Tom Nation, Dr. David Follows, Vikash Hemnath Seeboo, CC0, via Wikimedia Commons