More results...

Bragg's law is a simplistic model that has been developed to study crystal structures. In a crystal system, there are atoms are arranged in a periodic array. These atoms can scatter light and create a diffraction pattern.
Due to the diffraction of the different planes in the crystal, it will create a diffraction pattern that will give information about the atomic arrangement of the crystal. Bragg's equation helps to find the interspacing distance between the crystal planes.
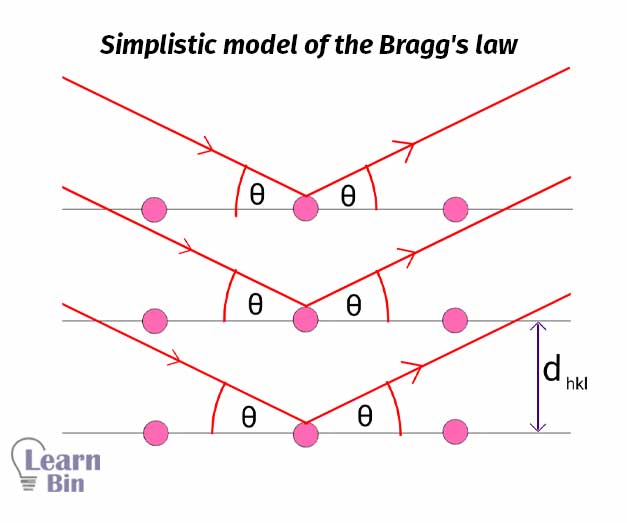
An X-ray beam is used as a coherent incident beam. When the X-ray hits the crystal plane, the atoms in the plane will scatter light. As shown in Figure 1, each plane will scatter the X-ray. If the Bragg's law is satisfied, a constructive interference is obtained. The inter-planer distance can be calculated using Bragg's equation.
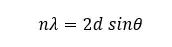
Where,
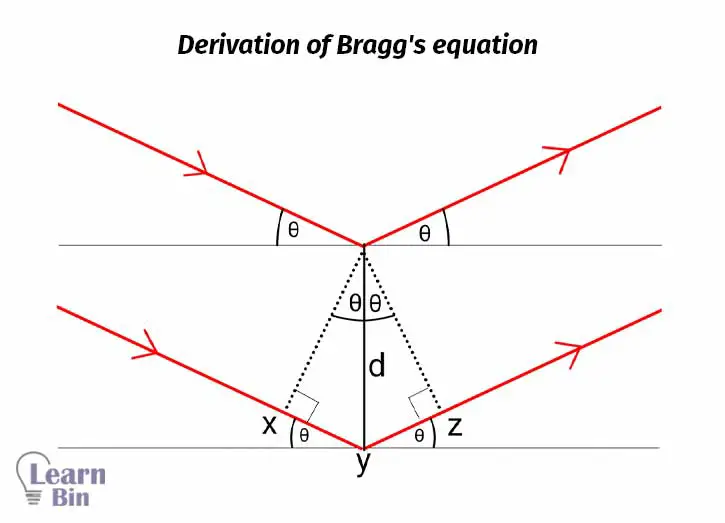
The X-ray will be diffracted by the atoms at the same angle as the incident beam. To obtain constructive interference, the scattered beams of each plane should be in the same phase. Let's take the scattered rays from the first plane and the plane beneath that plane. The extra distance traveled by the ray that scattered from the lower plane can be calculated as follows,
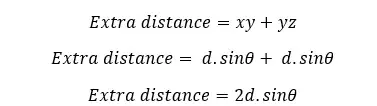
This extra distance can also be expressed in terms of the wavelength of the X-ray.
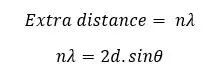
The value of n can be 1, 2, 3,… If it is a first-order diffraction, n is 1.
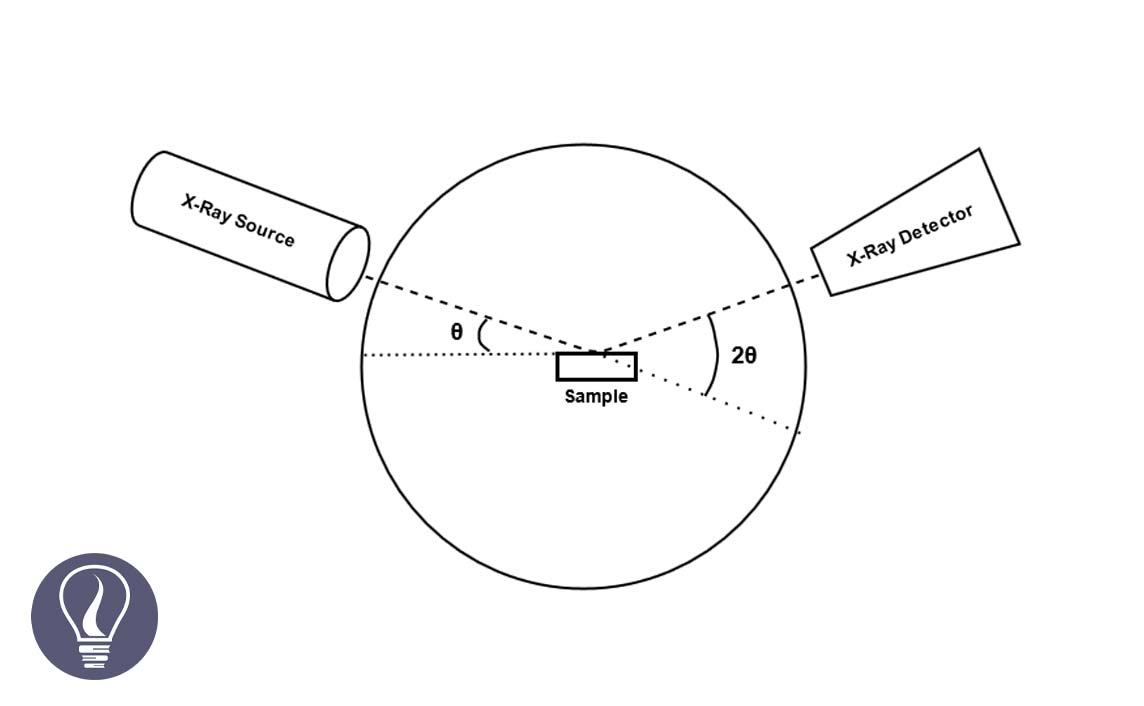
The X-ray powder diffraction method is typically used to analyze the crystal structures. Bragg's law is used in powder diffractometer.

The cover image was designed using an image by Tanuj Gupta, licensed under CC BY-SA 4.0, via Wikimedia Commons