More results...


The amount of reaction that has occurred when a system has reached equilibrium is called the equilibrium point. This varies from reaction to reaction. Factors affecting the equilibrium point are concentration, temperature, and pressure.
When the concentration of one or more reactants in a system at equilibrium is increased, the equilibrium of the system is disturbed. Then the forward reaction takes place more and the reactants become products. Now the system is back to equilibrium. Also, if the concentration of reactants is reduced, the products become reactants by performing the reverse reaction and the system becomes balanced.
Likewise, when the concentration of the product is increased, the reaction is directed in the direction of the reactant, making the reverse reaction take place more. The forward reaction takes place more when the product concentration is reduced. That is, the reaction is directed toward the product. Thus, the system returns to equilibrium.
When FeCl3 and NH4SCN are mixed, [Fe(SCN)]2+ complex is formed. This complex is a blood-red color.
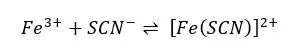
The effect of concentration on the equilibrium of the above system can be studied as follows. First, the resulting aqueous solution is diluted until clear and divided into four parts. One sample is taken as a control (reference) and an amount of FeCl3, NH4SCN, and NaOH are added respectively to the other three samples. The color of each system is compared with the reference.
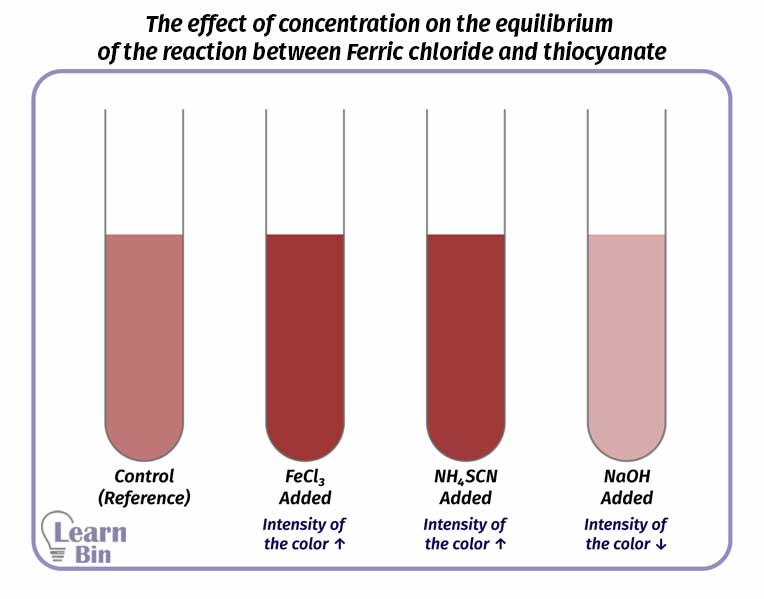
| Solution added | Observation |
| FeCl3 | The intensity of the red color increases |
| NH4SCN | The intensity of the red color increases |
| NaOH | The intensity of the red color decreases |
It can be observed that the red color is increased in the samples which are added FeCl3, NH4SCN. That is because more Fe(SCN) has formed. That is because the forward reaction has increased. According to Le Chatelier’s principle, when increasing the concentration of reactants, the reaction is forwarded in the product’s direction.
When the sample is added NaOH, it will form Fe(OH)3 precipitate. So, Fe3+ is removed from the medium. It can be observed that the intensity of the red color decreases. That is because the reverse reaction has increased. when the concentration of reactants decreases.
This system is in the following equilibrium,
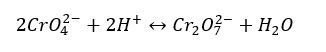
The chromate ion (CrO42-) is yellow in color and the Dichromate ion (Cr2O72-) is orange in color. When an acid is added to the above system, it can be observed that the system becomes orange color. That means the forward reaction has increased and more dichromate ions have been formed. Because the concentration of reactants has been increased.
When a base is added to the system, OH- ions react with H+ ions and form water (H2O). the concentration of products is increased. So, the system turns into a yellow color. That is because the reverse reaction has increased.
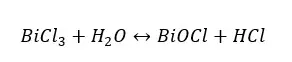
BiOCl is a white color precipitate. When the water is added to the above system, the amount of white precipitate is increased. When it is added HCl, the amount of the white precipitate is decreasing. Thus, the forward reaction is more favorable when adding reactants and the reverse reaction is more favorable when adding products.
Let’s consider the following reaction. We take the forward reaction as exothermic and the reverse reaction as endothermic.
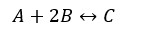
When the temperature of the above equilibrium system is increased, the equilibrium is disrupted. To reduce (somewhat) the increasing temperature, the reaction favors the endothermic direction (heat absorbing direction). That means the reverse reaction is increasing. Then the system became equilibrium again.
When the temperature of the above equilibrium system is decreased, the equilibrium is disrupted. To increase (somewhat) the decreasing temperature, the reaction favors the exothermic direction (heat-releasing direction). That means the forward reaction is increasing. Then the system became equilibrium again.
In an equilibrium system, when the temperature is increased the endothermic reaction is more favorable. When the temperature is decreased, the exothermic reaction is more favorable. By increasing or decreasing the forward and reverse reactions, the system became a new equilibrium.
The NO2/N2O4 system is in the following equilibrium.
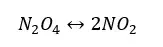
N2O4 is a colorless gas and NO2 gas is red-brown. Also, the forward reaction is endothermic, and the reverse reaction is exothermic. The effect of temperature on the above system can be observed as follows,
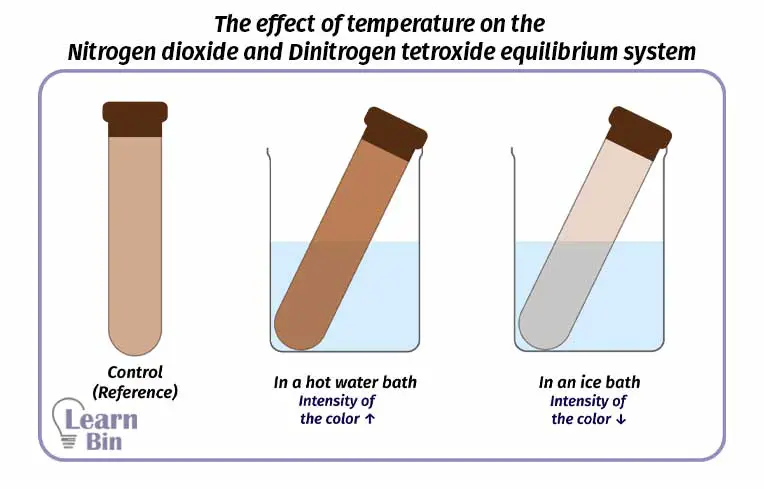
Three samples of NO2 gas with the same intensity of color are taken to three boiling tubes. Samples are labeled as A, B, and C.
| Sample | |
| A | Reference |
| B | Kept in an ice bath |
| C | Kept in a hot water bath |
After a while, it can be observed that the intensity of the red-brown color is decreased in sample B compared to the reference. This is because the reverse reaction has increased and more N2O4 has formed. According to Le Chatelier’s principle, the reaction has forwarded into an exothermic direction when the temperature is decreased.
As the same, it can be observed that the intensity of the red-brown color is decreased in sample C compared to the reference. This is because the forward reaction has increased and more NO2 has formed. According to Le Chatelier’s principle, the reaction has forwarded into the endothermic direction when the temperature is increased.
There are two ways to increase the pressure of a gas.
Here we consider the increase of pressure by decreasing the volume. Let’s consider the following equilibrium,
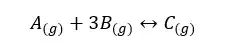
Generally, the amount of molecules is increased, and there are more collisions between the molecules. That caused to increase in the pressure of the system. As the same when the number of molecules decreases, the pressure also decreases. In the above system, there are four molecules in the reactant’s direction and there is only one molecule in the product direction. If the forward reaction is more favored, the pressure of the system is decreased. If the reverse reaction is more favored the pressure in the system is increased. Then the system becomes a new equilibrium.
Pressure affects only for gaseous system. If the number of molecules is equal on both sides of the reaction, such systems are not affected by the pressure. If there are liquids or solids on each side, those are not affected by pressure.
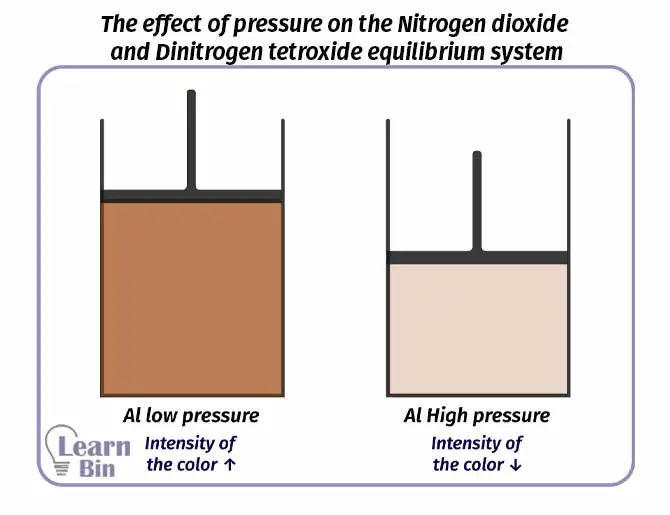
N2O4 /NO2 system can be used to study the effect of pressure. When the pressure is increased, the reverse reaction is a more favored one. When the pressure is decreased the forward reaction is a more favored one.
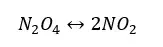
A sample of NO2 gas is taken. The volume of the sample is decreased and increased using a piston. When the volume is decreased (the pressure is increased), the intensity of the red-brown color of the sample decreases. That means the amount of NO2 has decreased and the reverse reaction has occurred more.
When the volume is increased (the pressure is decreased), it can be observed that the intensity of the red-brown color of the sample is increasing. That means the amount of N2O4 has decreased and more NO2 has formed. So, the forward reaction has occurred more.

The cover image was created using an image by Chokniti Khongchum from Pexels