More results...


Natural rubber contains a variety of metal cations such as Potassium, Sodium, Calcium, Magnesium, Iron, Copper, and Manganese. Measurement and control of metal ion content in natural rubber latex are highly significant in the NR industry. Because the ions in the latex directly affect the stability of the latex. Generally, ammonia is added to the latex to reduce the metal ion content. Ammonia will form a complex with metal ions and reduce the free ion concentration.
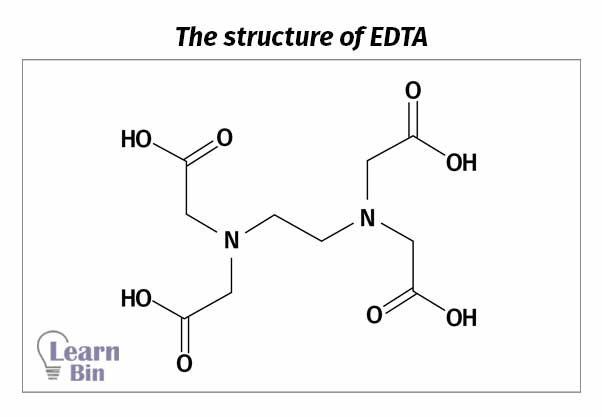
The methodology described below describes a titrimetric method to determine Zinc, Calcium, and Magnesium ion contents present in rubber compounds. These three ions can be estimated by titrating aliquots of an extract of the latex serum with EDTA (Ethylenediaminetetraacetic acid) under various conditions.
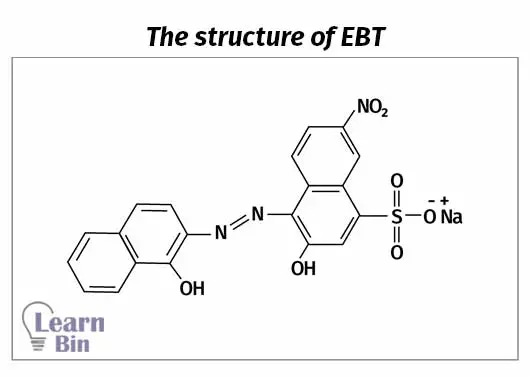
At pH 10 and using 'Eriochrome black T (EBT)' as the indicator, all the metal ions in the sample will react. At pH 10 with EBT and Potassium Cyanide (KCN) present, only the Magnesium and Calcium ions will react. Because Zinc ions will form a complex with cyanide.
At pH 12 Potassium cyanide and Murexide indicator, only Magnesium ions will be determined. From the results of the above three titrations’ concentrations of each ion can be determined.
Nearly 10 grams of latex are weighed into a clean dry beaker. To that beaker containing latex, 2% acetic acid was added to coagulate. Then the coagulum is collected using a glass rod. The coagulate is embedded and squeezed by a glass rod and washed. The total serum is collected along with washing with distilled water. The filtrate and washings are made up to 250 ml by adding distilled water in a graduated flask. This solution is used for the estimation of the metal present.
An amount of 25 ml of the test solution is pipetted out into a titration flask and it is added 5 ml of buffer solution. The sample is diluted up to 100 ml and it is added 5 drops of EBT indicator. This solution is titrated against 0.005 M EDTA solution to a clear-blue endpoint. Titration should be repeated until two similar endpoints are obtained.
An amount of 25 ml of test solution is added to 5 ml of buffer solution and 5 ml of KCN solution. This sample is diluted up to 100 ml and titrated against 0.005 M EDTA solution to a sky-blue endpoint using the EBT indicator. Titration is repeated until a similar endpoint is obtained.
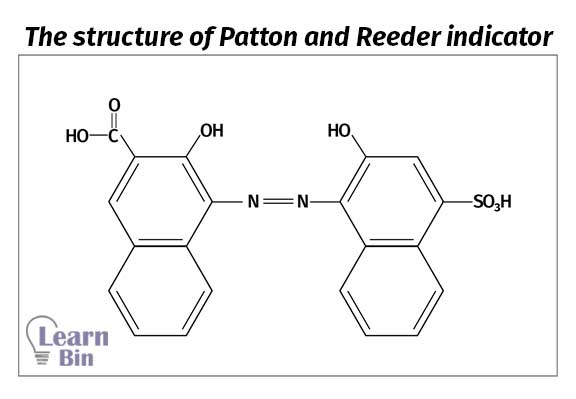
An amount of 25 ml of test solution is added to 5 ml of KCN and 5 ml of 8M KOH solution. The solution is diluted u to 100 ml, and it is added 0.1g of Patton & Reeder (P&R) indicator (Calconcarboxylic acid). Then the sample is titrated against 0.005 M EDTA solution to a red-violet endpoint. Titration is repeated until a similar endpoint is obtained.
Let’s assume that the following results have been obtained from the above titrations.
| Titration number | The volume of the test solution (cm3) | The volume of buffer solution (cm3) | The volume of EDTA (cm3) |
| 1 | 25.00 | 5.0 | 1.60 |
| 2 | 25.00 | 5.0 | 1.50 |
| Titration number | The volume of the test solution (cm3) | The volume of buffer solution (cm3) | The volume of KCN (cm3) | The volume of EDTA (cm3) |
| 1 | 25.00 | 5.0 | 5.0 | 1.60 |
| 2 | 25.00 | 5.0 | 5.0 | 1.50 |
| Titration number | The volume of the test solution (cm3) | The volume of buffer solution (cm3) | The volume of KCN (cm3) | The volume of KOH (cm3) | The volume of EDTA (cm3) |
| 1 | 25.00 | 5.0 | 5.0 | 5.0 | 1.60 |
| 2 | 25.00 | 5.0 | 5.0 | 5.0 | 1.50 |
Metal ion percentages of the sample can be calculated as follows.
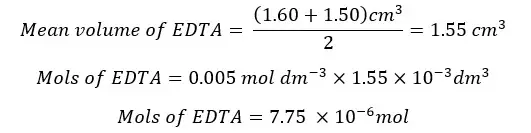
Each Mg2+, Ca2+, and Zn2+ react with EDTA 1:1 stereochemistry. Therefore, the mols of EDTA are equal to the mols of total metal ions.
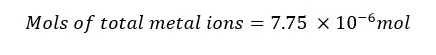
The total metal ions present in the 25 ml of the test solution is 7.75 * 10-6 mol. The mols of metal ions present in 250 ml of the test solution will be,
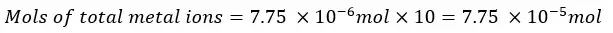
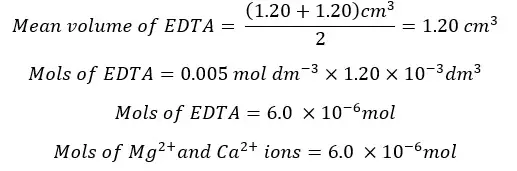
The Mg2+ and Ca2+ ions present in the 25 ml of the test solution are 6.0 × 10-6 mol. The mols of Mg2+ and Ca2+ ions present in 250 ml of the test solution will be,
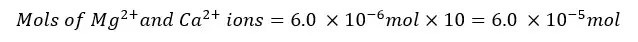
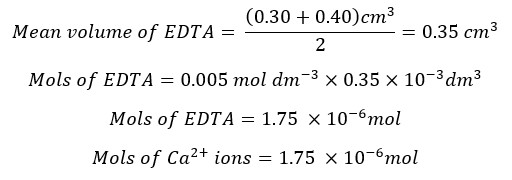
The Ca2+ ions present in the 25 ml of the test solution is 1.75 × 10-6 mol. The mols of Ca2+ ions present in 250 ml of the test solution will be,
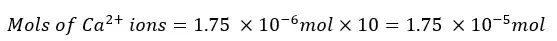
| Metal ions | The amount present in 25 ml of test solution |
| Ca2+, Mg2+, Zn2+ | 7.75 * 10-5 mol |
| Ca2+, Mg2+ | 6.0 * 10-5 mol |
| Ca2+ | 1.75 * 10-5 mol |
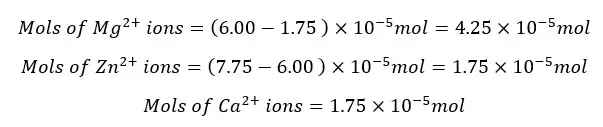
According to the above results, mass percentages of each metal in the initial latex sample can be calculated as follows.
Ca2+ percentage
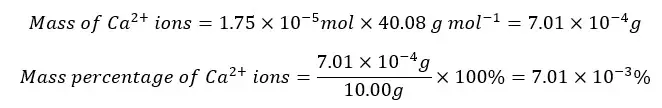
Mg2+ percentage
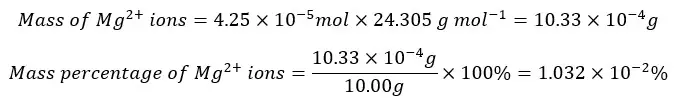
Zn2+ percentage
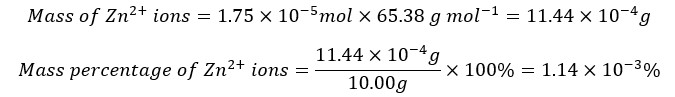
In fresh NR latex, rubber particles are surrounded by minus charges. Due to these minus charges, rubber particles cannot come to each other. They are repulsed by each other. But when adding acetic acid to the latex, minus charges are neutralized. Therefore, rubber particles can interact with each other and they will coagulate.
As mentioned above rubber particles are surrounded by minus charges. Divalent metal ions like Ca2+, Mg2+, and Zn2+ can bond with these minus-charged sites. Since these ions are divalent, one ion can bond with two minus-charged sites. One metal ion acts as a bridge that interconnects two rubber particles. So, when metal ions are present in latex, the latex will be coagulated.
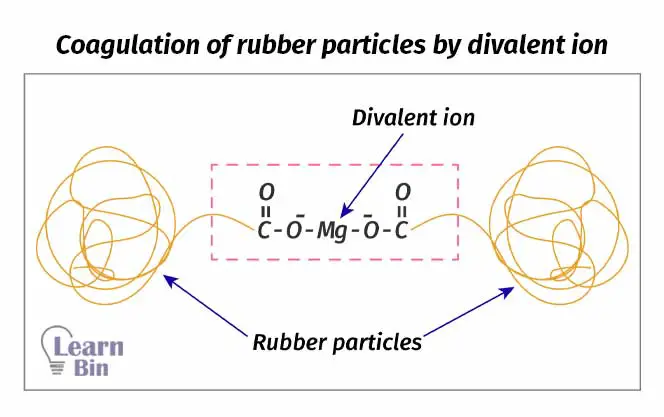
EDTA is a versatile chelating agent. It can form four or six bonds with a metal ion and it chelates with both transition metal ions and main group metal ions.
A blue dye called Eriochrome Black T is used as the indicator in 1st and 2nd titrations. This indicator forms a complex with Mg2+ ions and produces a pink/purple solution. The indicator is blue when it is not complexed with Mg2+ and the solution is basic.
When titration with EDTA it reacts with divalent ions (Ca2+ and Zn2+). Once all the divalent ions in the solutions have reacted with EDTA, it starts to react with Mg2+ ions that are complex with the EBT indicator. Therefore, the solution becomes blue at the endpoint.
A blue dye called Patton & Reeder (P&R) has been used as the indicator in titration 3. P&R indicator forms a complex with the Ca2+ ions and turns the medium from blue color to pink. When titration with EDTA, Ca2+ ions form a complex with EDTA. Because the EDTA-Ca2+ complex is more stable than the P&R-Ca2+ complex. As Ca2+ ions are de-complex (P&R-Ca2+ complex decomposed) with the indicator, the solution becomes blue again.
When adding KCN, CN- ions form a strong complex with Zn2+ ions. This complex is stronger than the Zn-EDTA complex. Therefore, when titrating, only Ca2+ and Mg2+ ions will react with EDTA.
In titration 3, both KCN and KOH are added. CN- forms a strong complex with Zn2+ and OH- will react with Mg2+ forming Mg(OH)2. Therefore, in titration 3, only Ca2+ ions will react with EDTA.

The cover image was created using an image by Vyacheslav Argenberg, licensed under CC BY 4.0, via Wikimedia Commons