More results...


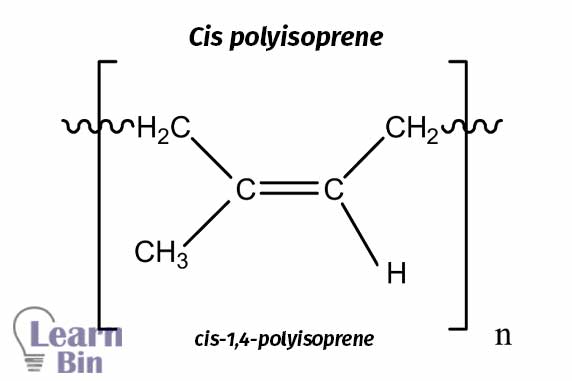
Natural rubber is a naturally occurring latex with the long-chain polymer of cis polyisoprene. Natural latex is a white color fluid that is collected by tapping the rubber tree. There are different Natural rubber species more than 200. But Hevea brasiliensis is the commercially significant source of natural rubber.
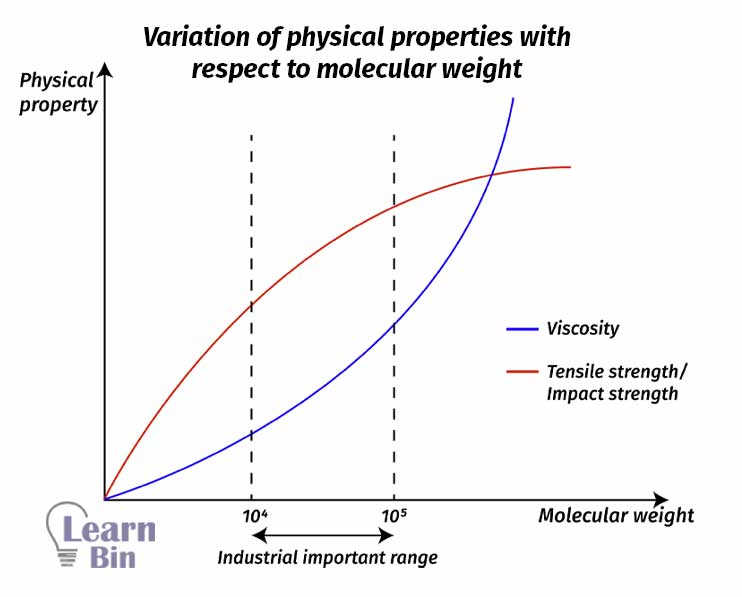
Natural rubber is a high molecular weight polymer. The molecular weight can be up to 6000000 g/mol. Generally, synthetic rubber has a molecular weight range between 5000 – 10000 g/mol. The molecular weight of natural cannot be controlled and the molecular weight of synthetic rubber can be controlled. Polymers with high molecular weight have high physical properties.
Because the polymer molecules are entangled and connected to each other well. Therefore, the intermolecular interactions are high. So, the physical properties such as tensile strength, impact strength, shear stress, viscosity are increasing. But the commercially important range of molecular weight is 10000 – 100000 g/mol.
Natural rubber (Cis polyisoprene) molecules are very nonpolar. Therefore, it cannot dissolve in polar solutions. However natural rubber is very difficult to dissolve in nonpolar solutions also. Because natural rubber has long-chain molecules, they are highly entangled. Vulcanized rubber does not dissolve in liquids either and it swells in solvents.
Polymers change their properties from a glassy state to a rubbery state when increasing the temperature. In the glassy state, the polymer is brittle, and it is hard to work with glassy polymers. The temperature at which polymer changes from a glassy state to a rubbery state is called “Glass transition temperature (Tg)”. The Tg of natural rubber is around -67 ℃ and the melting point is approximately 180 ℃. In this region, the natural rubber maintains its rubbery properties. So, the natural rubber is in a rubbery state in a high range of temperatures. After vulcanizing the Tg will increase.
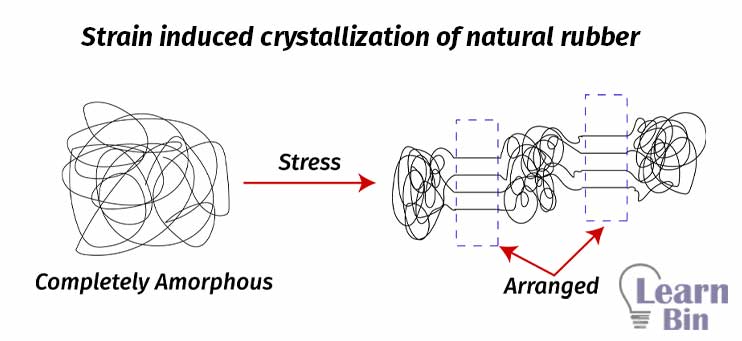
Natural rubber is an amorphous polymer. When it is applied stress, the rubber molecules are arranged along the direction of the force. Therefore, some regions of the rubber are crystalized. This phenomenon is called strain-induced crystallization. This is a unique property for natural rubber. With the crystallization, the Tg and melting temperature (Tm) also increased. As well as the tensile strength of the polymer will increase. Therefore, natural rubber shows high tensile properties even without fillers.
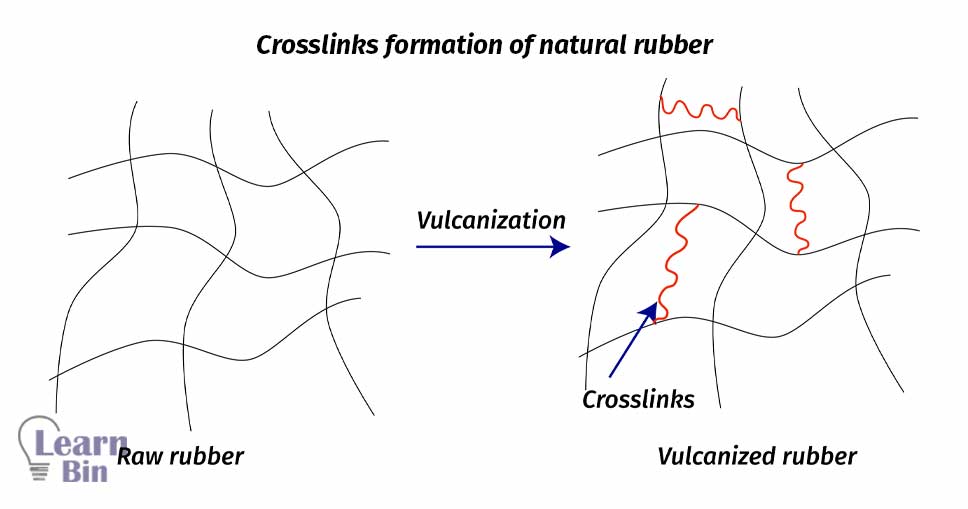
Vulcanization is the process that forms crosslinks between rubber chains. This will enhance the elastic properties of natural rubber. When un-vulcanized rubber (raw rubber) is applied a force, it will stretch, and then remove the force the rubber will not be completely recovered to its original dimensions. So, un-vulcanized rubber simply behaves like plastic. But the vulcanized rubber will stretch when a force is applied and will recover to its original state (or very close to its original state) when force is removed. This is because of the cross-links in vulcanized rubber. Vulcanized rubber is an example of elastomers.
| Property | Natural rubber |
| Density | 1100Kg/m3 |
| Hardness | 30- 90 shore A |
| Tensile strength | 5 MPa |
| Young’s modulus | 50 MPa |
| Elongation | 500% - 900% |
| Compression set | 10-30% |
| Specific gravity | 0.92-0.93 |
| Thermal conductivity | 0.5 W/mK |
| Thermal expansion coefficient | 220 ×10–6 m/mK) |
| Glass transition temperature (Tg) | –72 ℃ |
| Melting temperature (Tm) | 177 ℃ |
| Heat capacity | 1300 J/gK |

timcorubber.com - Natural Rubber: A Tough Material for Physically Demanding Applications
material-properties.org - Rubber – Density – Strength – Melting Point – Thermal Conductivity
The cover image was designed by using an image by Slashme, licensed under CC BY-SA 4.0, via Wikimedia Commons.
It's nice that you pointed out how natural rubber is a high molecular-weight polymer. I was watching a TV show yesterday and it showed how a factory produces custom rubber parts. Rubbers got very unique properties, which make them very useful for crafting parts.