More results...


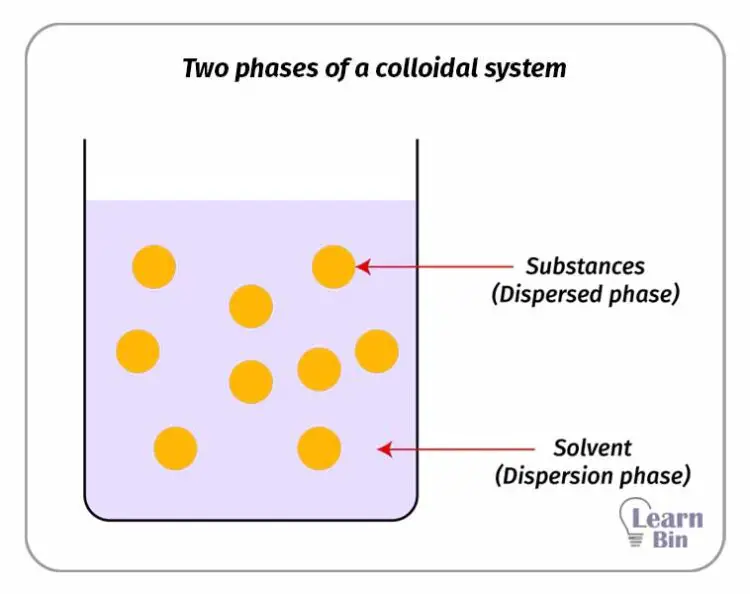
A colloidal system is a homogeneous non-crystalline system consisting of large molecules or ultra-microscopic particles (1-1000 nm). A colloidal system has two phases. One substance is dispersed in another substance.
| Colloidal system | Dispersed phase | Dispersion medium |
| Foam | Gas | Liquid |
| Solid foams | Gas | Solid |
| Aerosol | Liquid | Gas |
| Emulsion | Liquid | Liquid |
| Solid emulsions (gels) | Liquid | Solid |
| Smoke | Solid | Gas |
| Sol | Solid | Liquid |
| Solid sol | Solid | Solid |
In a colloidal solution, the dispersion medium is a liquid. Colloidal solutions have intermediate properties of true solutions and suspensions.
E.g.:–
A homogeneous mixture that consists of two or more substances, can be defined as a true solution. Those substances are dissolved in a solvent. The particle size of the substances is less than 1 nm. Particles in a true solution cannot be filtered.
E.g.:-
Suspension can be defined as a heterogeneous mixture of two or more substances. Those substances are dispersed in a solvent. The particle size of the substances is higher than 1000 nm. Sometimes the particles will settle down at the bottom. In a suspension, dispersed particles are visible to the naked eye.
E.g.:-

The liquid in a liquid system, liquid in a solid system, and solid in a liquid system are the most common types of colloids. A colloidal solution where a solid in a liquid is called 'sol'.
| The dispersion medium of a sol | Type of the sol |
| Water | Hydrosol |
| Alcohol | Alco-sol |
Sols can be categorized into two categories based on the affinity of the dispersed phase towards the dispersion medium.
Lyophilic sol is a colloid where the dispersed phase has a strong affinity to the dispersion medium.
Lyophobic sols are colloids where the dispersed phase has less affinity or no affinity to the dispersion medium.
| Property | Lyophilic | Lyophobic |
| Preparation | Can easily make by shaking or mixing. | Needs special machines/ mills to prepare |
| Recovery after precipitation | Easily recovered after precipitation. | Precipitation is irreversible. Once it is precipitated, it cannot be bought back to colloidal form. |
| Charge | The colloidal system can be positive, negative, or neutral. | The system is positive or negative. Cannot be neutral. |
| Stability | Very stable. The system is self-stabilized. | The system is unstable. It needs a little bit of electrolyte to make the system stable. |
| Electrophoresis | Dispersed particles can migrate to anode, cathode, or not migrate. | Dispersed particles migrate to the anode or cathode. |
| Hydration | Have very strong hydration. Solvent loving | Not going to hydrate very well. Solvent hating. |
| The concentration of the dispersed phase | Can have a higher concentration of dispersed phase. | Only low concentrations are available. |
| Property | Example |
| Optical properties | Tyndall effect |
| Kinetic properties | Brownian motion |
| Electrical properties | Electric charge Electrophoresis Precipitation Coagulation |
In a colloid or suspension, the light will be scattered by the dispersed particles. It will make beams of light visible. Therefore, the path of the light is visible. This effect is known as the 'Tyndall effect'.
The amount of scattering depends on the size of the dispersed particles and the wavelength of the light.

In true solutions, we cannot see the light path. There is no Tyndall effect in true solutions.
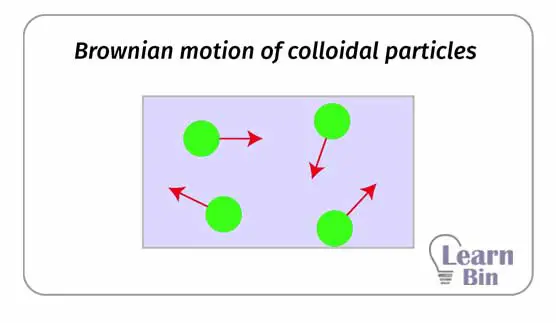
The continuous random motion of particles is known as the 'Brownian Motion'. In a colloidal solution, dispersed particles travel in several directions at a given time.
1D motion – particles are considered as moving only forward and backward in x direction.
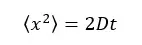
2D motion – particles are considered as moving on a plane.
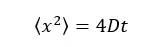
3D motion – particles are considered as moving in 3D space.
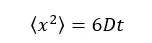
Where,
The diffusive coefficient of the system is given by the “Einstein – Stokes equation.”
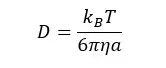
Where,
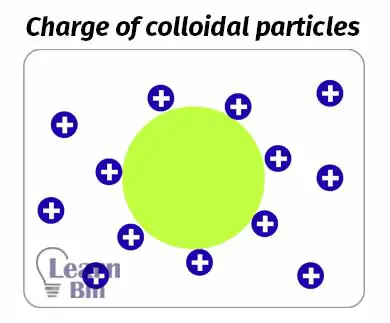
Most of the colloidal particles are carrying a positive or negative charge. If the wan-der Wal’s attractions of particles are much higher, particles are aggregated and settle down at the bottom. But in a colloidal system, there is another force to overcome the aggregation. It is the repulsive force between colloidal particles. This repulsive force is given by the charged particles in the colloidal system.
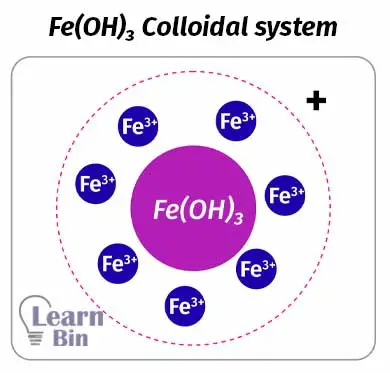
In an aqueous medium, FeCl3 will form a red-color colloidal solution of Fe(OH)3 as follows.
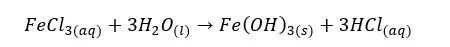
Fe(OH)3 will absorb Fe3+ ions in the system. Therefore, Fe(OH)3 is surrounded by positively charged ions. So, the colloidal particles are positively charged.
We can prepare positively or negatively charged colloidal particles by changing the way we prepare the colloid. Let’s consider the following colloidal system.
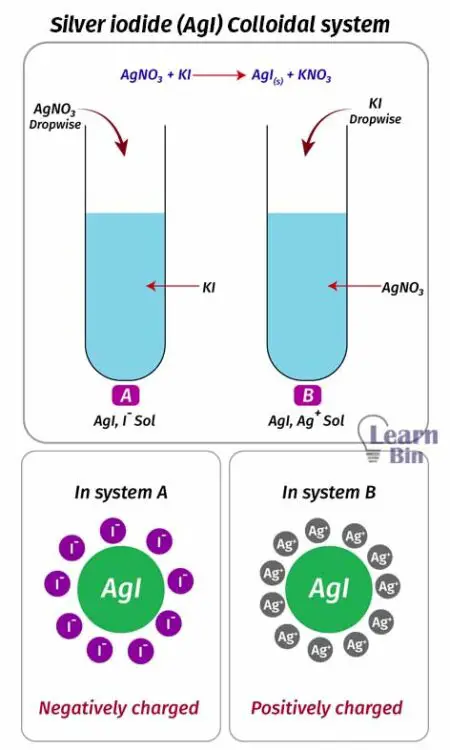
When we add AgNO3 solution dropwise to the KI solution, negatively charged colloidal particles will form. Because in the KI solution, there are excess amounts of negatively charged ions (I-). So, AgI will be surrounded by I- ions and form negatively charged colloidal particles.
But if we add KI solution dropwise to an AgNO3 solution, negatively charged colloidal particles will be created. Because there is an excess amount of positively charged ions (Ag+) in the AgNO3 solution. So, AgI is surrounded by Ag+ ions and forms positively charged colloidal particles.
When colloidal particles are surrounded by a layer of charged particles, that layer is also surrounded by an oppositely charged layer. Therefore, a colloid particle is surrounded by two charged layers. This double layer is known as the electrical double layer.
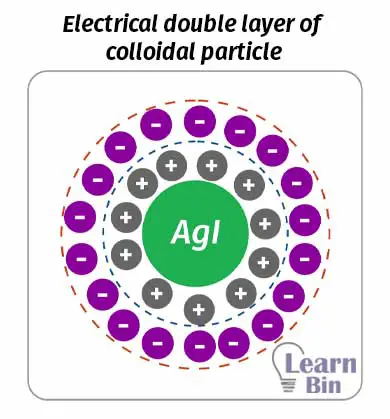
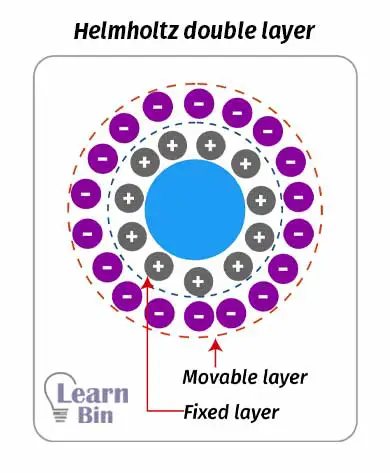
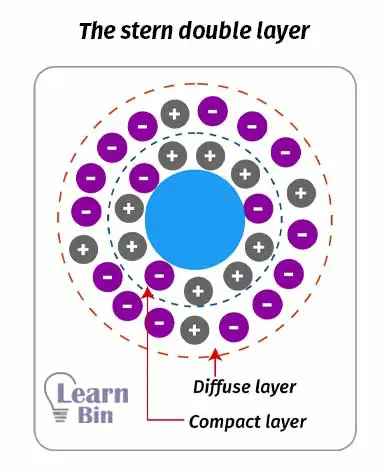
An electrical double layer is described in two models which are the “Helmholtz double layer” and “The stern double layer”. Helmholtz's double-layer model describes that there is a fixed charged layer surrounding the colloid particle. And there is an outer charged layer that is movable.
The stern double layer describes that there is a compact charged layer surrounding the colloid particle. Surrounding the compact layer there is a diffusive later. Both layers have positive and negatively charged particles. But the overall charge is negative or positive (not neutral).
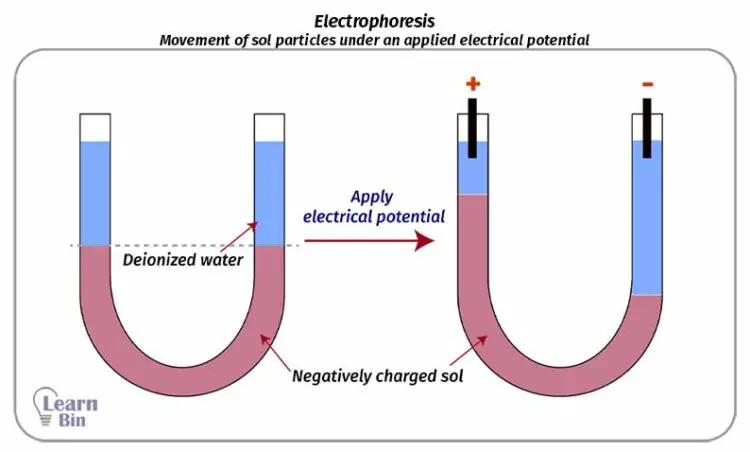
Electrophoresis is the movement of sol particles under an applied electrical potential. According to the charge of the colloidal particle, they will move to the positive electrode or negative electrode. Positively charged particles will move to the negative electrode and negatively charged particles will move to the positive electrode.
The aggregation and precipitation of discharged sol particles are known as coagulations or precipitation. To coagulate sol particles, the stability of the colloid should be broken down. The stability of colloids can be broken down by the four methods below.
When a positively or negatively charged colloid is added to an electrolyte, charged particles of the electrolyte will neutralize the electrical double layer of the colloidal particles. Therefore, the stability of the colloid will be collapsed. So, particles will be aggregated and coagulated.
A colloidal solution is inserted between positive and negative electrodes externally. Charged particles in the electric double layer travel to the electrodes according to their charge. Therefore, the stability of colloid collapse and particles will be coagulated.
When mixing oppositely charged sols will neutralize each other. And the particles will be coagulated.
When boiling, the sol is supplied with energy. Therefore, the collision between the sol particles and water molecules is increased. So, the collisions remove adsorbed charged particles. Then, the particles will coagulate.

The cover image was created using an image by Pixnio
Figure 03: Contains an image form PxHere