More results...


In addition to resins, solvents, and pigments, paint also contains additives. Typically, 0.01% - 1% of the volume of paint is additives. The main function of an additive is to prevent defects in paint and coating. The most common defects in paints are foam bubbles, poor leveling, flocculation, sedimentation, microbial growth, and discoloration. There are several classes of paint additives.
Generally, foams occur when applying the paint. Liquid foams are a fine distribution of a gas in a liquid. Foam bubbles occur due to the interfacial tension differences between the pain and the substrate.
Defoamers are liquids with low surface tension and will prevent foam formation. Defoamers penetrate the foam lamellae and spread across the surface. this will create interfacial tension difference which destabilizes the lamellae and cause the foams to collapse.
Generally, surfactants or polymeric substances are added as dispersing agents. The functions of dispersing agents are to spread the pigment particles in the solvent evenly and stabilize them.
Common defects in paints are poor substrate wetting, floating, poor leveling, orange peel, etc. the main reason behind all the above defects is known as the differences in surface tension of materials. It leads to the formation of surface defects in the paint film. Surface additives are added to the paint to minimize the surface tension differences. Generally, surface additives are polysiloxanes (silicones) and polyacrylates (acrylate additives).
The rheology of paint can be described by its viscosity. Rheology additives are employed to modify the flow behavior of paint.
After the paint is applied on a surface drying process is occurred physically and/or chemically. Physical methods are evaporation of the organic solvent, evaporation of water, or cooling the polymer melt. The chemical method is cross-linking. Low molecular products will react with low or medium-molecular weight binder components.
Higher crosslinking results in better drying. If the binder components have a higher degree of unsaturation, it will result in more crosslinks. But in some binders’ the degree of unsaturation is low. Therefore, driers are added.
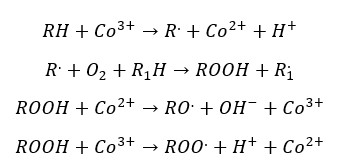
Paints and paint films are easily attacked by microorganisms. Microbial growth in a liquid paint cause gas formation, discoloration, bad odor, etc. To prevent microbial activities biocides and fungicides are added to the paint.
UV light causes for degradation of polymers. This will result in loss of gloss, crack formation, and discoloration. Therefore, light stabilizers are required. There are two types of UV stabilizers according to the mechanism of stabilization
Corrosion inhibitors prevent the corrosion of the substrate. Zinc dust is commonly used as a corrosion inhibitor. It will give the iron surface cathodic protection.

The cover image was created using an image by Delcea Nicolae Cosmin from Pexels