More results...


Pigments are colorful compounds that give color to other materials. Pigments that are used in the paint industry, are natural or synthetic and organic or inorganic. The functions of pigments in paint are,
Tinting strength is a measurement of its opacity. It gives an idea of how much pigment is required to get a particular color. If a lot of pigment is required to get the desired color, tinting strength is poor. Pigments with higher tinting strength do not change their color significantly when it is added to white color pigments. Sometimes the tinting strength can be increased by blending the pigment into a fine powder.
Lightfastness is the resistance to changing color (fading) when exposed to light.
Hiding power is the ability to hide the substrate. It depends on the wavelength of the light, the ability of the film to absorb and scatter light, the refractive index of the pigment, the particle size of the pigment, etc. Pigments with high light absorption coefficients show higher hiding power.
White paint pigments like Titanium dioxide get their hiding power from their ability to scatter light. Pigments with a higher refractive index have higher scattering.
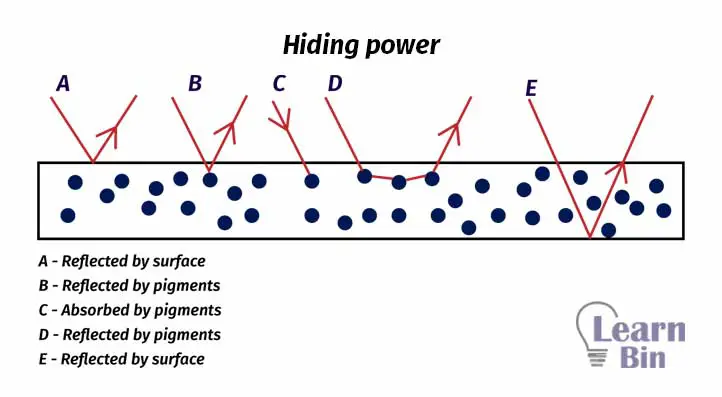
The scattering of light depends on the size of the pigment particles. Particle size should be higher than the wavelength of light to scatter the light.
| Pigment | Particle size (μm) |
| Iron blue | 0.01 – 0.2 |
| Titanium dioxide | 0.02 – 0.3 |
| Red iron oxide | 0.3 – 0.4 |
| Natural crystalline silica. (White color) | 1.5 – 9 |
| Strontium chromate (SrCrO4) – Yellow color | 0.3 - 20 |
The shape of the paint pigments determines the properties of the paints like flow properties, setting properties, barrier properties, the durability of the paint film, etc. based on the crystallographic structure, pigments can be divided into three categories.
Rod shape pigments will poke through the paint film. This will reduce the gloss of the paint. Needle shape pigments are crack initiators. They will cause cracks in the paint film. Platelets or leaf-like pigments can arrange parallel to the substrate. So, it will prevent the substrate from moisture, oils, radiation, etc. normally pigments with rough surfaces are used in undercoats.
The ratio of the weight of the pigments to its net volume.
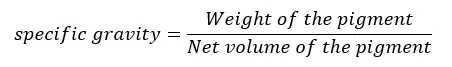
Colored pigments improve the appearance and provide color to the paint. It depends on the particle size, shape, and distribution of the pigment.
Hiding pigments provide opacity to the paint film. Most of the time, hiding pigments are carbon black or Titanium dioxide (TiO2)
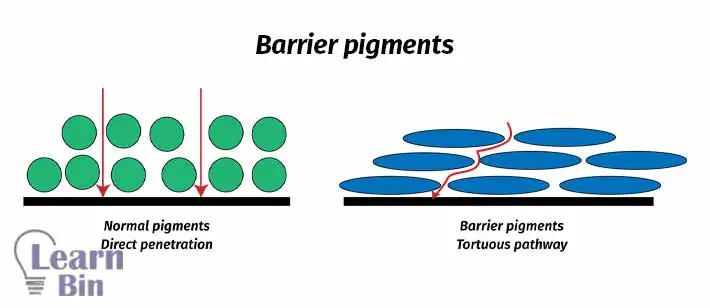
Barrier pigments are sheet-like micro-size fillers. Their flat surfaces are settled parallel to the air–polymer (binder) surface. Therefore, water and Oxygen molecules cannot penetrate directly into the substrate. Those molecules have to flow through tortuous pathways.
Reinforcing pigments are commonly used in the rubber industry. They give additional strength. The most common reinforcing pigment is carbon black.
Extender pigments are added to the pigments to increase the pigment volume. So, it causes to reduce the cost of the pigment.
e.g., silica, Calcium carbonate (CaCO3), Barium sulfate
Sacrificial pigments are used in anticorrosive paints. Pigments sacrifice themselves to protect iron substrate (Cathodic protection). Sacrificial pigments contain metals that are higher in the electromotive series than the metal of the substrate to be protected.
Without anticorrosive paint, the iron surface will corrode as follows,
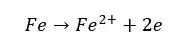
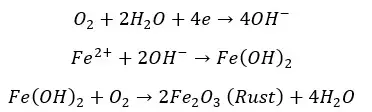
Under corrosion conditions, the sacrificial pigments are more reactive than the substrate. Therefore, the substrate will be the cathode and the pigments will be the anode. If Zinc based pigments are used for the iron substrate, the following reactions occur.
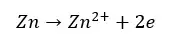
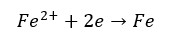
Inhibitive pigments reduce the rate of corrosion by forming a protective layer (a barrier layer) over the substrate. These inhibitive pigments release soluble species from the pigments to the solvent. They will penetrate the metal surface and form a protective layer. This is a passive way of corrosion inhibition.
e.g.,
Both pigments and dyes are aggregated in dry form. But when they dissolve in a solvent, dyes make a transparent solution where pigments give their color and opacity to the solvent.

The cover image was created using an image by André Zivic from Pixabay