More results...


PMMA -Polymethyl Methacrylate (C₅O₂H₈) n (plexiglass) is an engineering thermoplastic that is synthesized by the polymerization of Methyl Methacrylate.
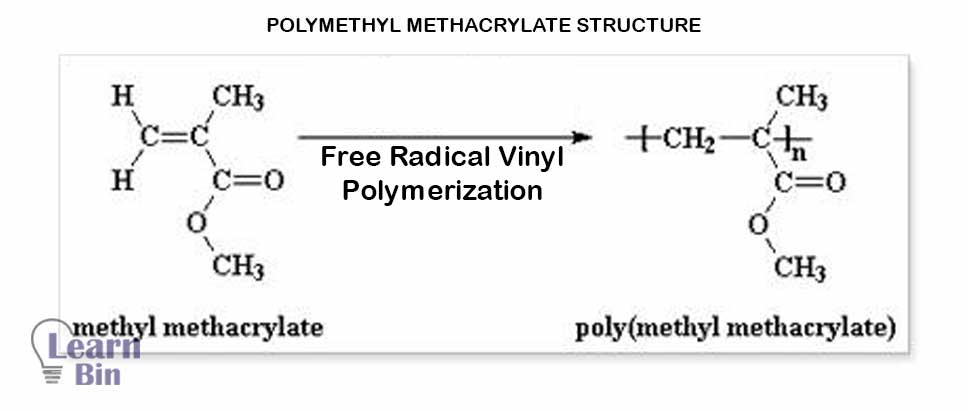
Methyl methacrylate will polymerize readily during monomer storage. Polymerization may start under light, heat, presence of peroxide. In the commercial production process, the monomer-methyl methacrylate is supplied combined with an inhibitor like hydroquinone (up to 0.10%). Hydroquinone is removed before polymerization by distillation under pressure or, in some cases, by rinsing with an alkaline solution.
Hydroquinone is an aromatic organic compound also known as benzene-1,4-diol or quinol. It is a type of phenol, a derivative of benzene. The chemical formula of Hydroquinone is C6H4(OH)2.
Methyl methacrylate monomer is polymerized by Free-radical polymerization in a fully closed cell or chamber free of oxygen or blanketing the monomer with an inert gas. The reaction chamber should be Oxygen-free because the presence of oxygen will affect the rate of reaction owing to the formation of methacrylate peroxides in a side reaction. azo-di iso-butyronitrile is added as an initiator. The temperature of the chamber is about 100°C.
Polymerization can be done by the Bulk process, Emulsion process, Suspension process, or Solution process. But generally, the polymerization of Methyl methacrylate is done by the bulk polymerization process.
Bulk polymerization is extensively used in the manufacture of sheets and to a lesser extent rods and tubes. Bulk polymerization is a polymerization process that only involves monomers and initiators as the main components. This process does not use any solvent. The monomer is used as the reaction medium, and the initiator is dissolved in the monomer.
This process is exothermic. The reaction medium becomes more viscous because no solvent is used. The acceleration effect happens with polymers that are not soluble in their monomers. Due to the high viscosity of the medium, the radical end becomes entrapped in the polymer, and termination reactions become very difficult.
Methyl methacrylate monomer is a poor solvent for polymethyl methacrylate polymer. It causes growing radicals to coil while in solution. The termination reaction is determined by the speed at which the radical ends reach the surface of the coil and is therefore available for mutual termination.
Bulk polymerization of methyl methacrylate includes 2 stages:
Pre-polymer is a partially polymerized monomer. It has an intermediate molecular mass of the polymer. This pre-polymer can be polymerized further.
The first step of the pre-polymerization of methyl methacrylate is removing the inhibitor from the monomer.
Then methyl methacrylate monomer is mixed with 0.5% azo-di iso-butyronitrile (initiator) and lets the reaction happen for 8 min at T=90℃. Then, the prepolymer is added to other additives such as plasticizers, pigments, and UV light absorbers. The oxygen that is dissolved in the prepolymer is removed by vacuum application. The final prepolymer is in a liquid state. This syrup is cooled up to room temperature and stored under refrigeration.
The mold of cast acrylic sheet is typically made of large tempered glasses with a separating gasket around the edges. First, the mold is assembled by two large tempered glass plates which are separated to produce the desired thickness of the sheet. Then, the mold is sealed by a gasket at the edge.
For each sheet, a separate mold is constructed. Then the mold is poured into the acrylic syrup (prepolymer) by a funnel or other device. pouring the syrup into a casting call. In the “water bath technique,” the sealed molds are submerged in a water bath which maintains and controls the curing temperature. If not, molds are led through a heating tunnel.
The time it takes to cross a tunnel in a typical system is about 16 hours. For the first 14 hours, the cell (mold) passes through heating zones at about 40°C. at this first heating zone, a slow polymerization reaction occurs. This prevents the monomer from boiling.
After 15 hours (total time) the cell is passed through the second heating zone. In this zone, the temperature is increased up to 97℃, at which temperature it is held for a further half-hour. Then the mold is cooled and the formed sheet from the mold is removed.
This process manufactures 100% virgin PMMA with a density of 1.2g/cm3. The thickness range is between 4-200mm and the main dimensions of a sheet are 1220x2440/1220x1830mm.
To make solid bars and rods, vertical aluminum tubes or other shape cylinders are used as the mold. Molds are filled with syrup and slowly dipped into a water bath at 40°C. The mold can be easily removed due to the natural shrinkage of the material.

Polymethyl-methacrylate is a clear and optically transparent thermoplastic. The amorphous nature of PMMA gives the transparent nature itself.
The presence of the pendant methyl (CH3) groups prevents the polymer chain from packing in a crystalline manner and rotating freely around the carbon-carbon bond. Therefore, PMMA tends to be amorphous in nature. Microscopic and X-ray analyses generally indicate that PMMA is amorphous. PMMA is considered about 54% syndiotactic, 37% tactic, and 9% isotactic.
Polymethyl methacrylate absorbs a small amount of light. But for normal incident light, it shows about 4% reflection at each polymer-air interface. The transmission of normal light through a parallel sheet of PMMA is free from any deformations. That means it is transparent.
PMMA is a polar thermoplastic material, so mechanical, electrical, and other properties are strongly dependent on temperature and humidity.
The glass transition temperature of PMMA is 114℃. The amorphous density of the material at 25℃ is 1.17 g/cm3. The molecular weight of the repeat unit is 100.12 g/mol.
In terms of impact resistance, it is superior to untreated glass, and although it does crack, any parts made are less sharp and jagged than glass and are generally less harmful.
For low-frequency tasks, PMMA is a good electrical insulator. But, especially at high frequencies is inferior to polymers such as polyethylene and polystyrene.
PMMA is not attacked by alcohol alone but alcohol with carbon tetrachloride and ether causes swelling.
Dissolved by most aromatic and chlorinated hydrocarbons.
Water absorption of PMMA is low but can have a significant effect on dimensions and less impact on mechanical properties.
Polymers are blended with additives to improve or build new properties. The most important additives are pigment and dyes. These pigments and dyes should be stable to both processing and service conditions. Generally, they should not affect the polymerization reaction when used in castings and they should have good weathering resistance.
Plasticizers are added to the polymer to make the material softer and more flexible. It will enhance the melt flow. But sometimes plasticizers will reduce the mechanical properties of the finished product.
Ultraviolet absorbers (UV absorbers) are added to the material to improve the light stability of the material. Some examples of UV stabilizers are shown below.
The standard material for automobile rear lamp housings is Polymethyl methacrylate. Factories and offices frequently use PMMA housings. Other applications of PMMA are,


PMMA - Polymer Science Learning Center
Cover Image by Edinaldo Maciel edy boy from Pixabay