More results...


The majority of the organic molecules and functional groups are transparent to ultraviolet (UV) and visible light. UV Visible spectroscopy (ultraviolet-visible spectroscopy) uses this electromagnetic radiation ranges to generate a spectrum and gain other information about molecules. UV-Vis spectroscopy has more limited applications when compared to spectroscopic methods like infrared or NMR (H-NMR, 13C-NMR). UV Visible spectroscopy is mostly applied in analyzing organic compounds.
So, molecules have the ability to absorb those transmitted radiations from those wavelengths and use them to excite their electrons to a higher energy level from the ground state. By detecting the absorbance or the transmittance at different wavelengths, details/ information about the molecule and the solution can be gained.
For example, these data can be used to calculate the concentration of a solution or get an idea about the structure of the molecule. Also, if these results were combined with more detailed spectra like Infrared or NMR, can get very valuable information about the structure of the molecule.
When radiation (which contains a range of wavelengths) is transmitted through a transparent material/ molecule, a portion of that radiation can be absorbed by the material. If the transmitted radiation (transmitted beam) passes through a prism, a spectrum can be observed. This is called as “Absorption spectrum”. That spectrum contains a gap because the material/ molecule has absorbed a specific wavelength from the incident beam.
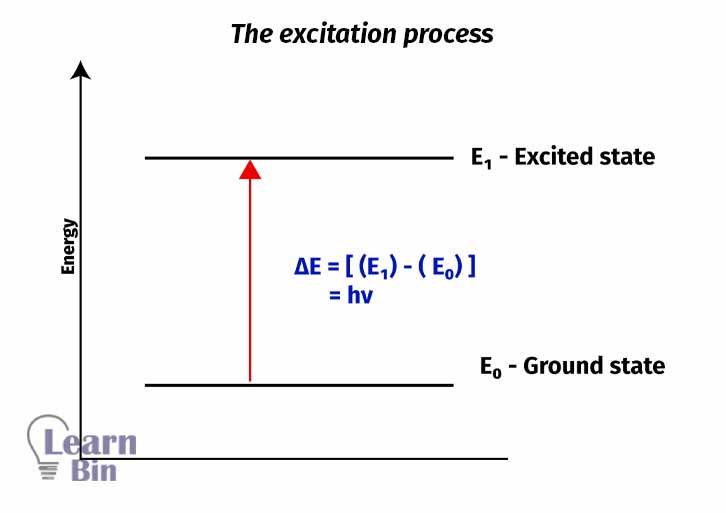
Molecules or atoms use that absorbed energy to excite their electrons to an excited state (higher energy level) from the ground state (lower energy level). The energy difference between the ground and the excited states is exactly equal to the amount of radiation absorbed.
In UV-Vis spectroscopy, radiation from ultraviolet and visible light excites electrons of the molecules from a ground state to an excited state. In this process, electrons were excited from the occupied molecular orbital to the unoccupied molecular orbital.
Normally the energy difference between electronic energy levels in most of the molecules is ranges from 125 kJ/mol to 650 kJ/mol.
The outermost energy level called the “valance shell” is the only energy level that participates in electronic excitation and bonding. So that energy level is the only energy level considered in molecular orbital theory.
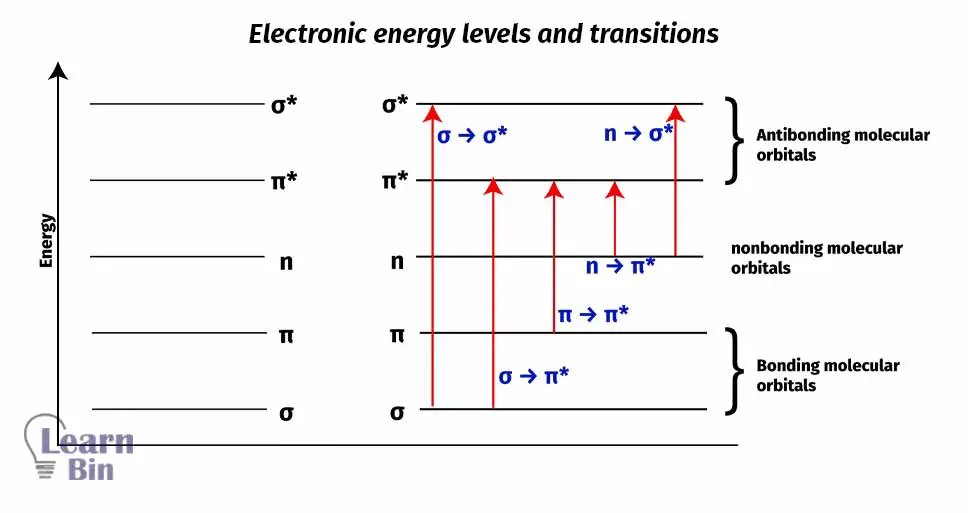
Normally, the most possible transition happens between “Highest occupied molecular orbital” (HOMO) to “Lowest unoccupied molecular orbital” (LUMO). LUMO has greater potential energy than "HOMO".
Generally, molecules contain 3 types of molecular orbitals. (Please take a look at figure 04)
The σ orbitals correspond to σ bonds. The π orbitals correspond to π bonds. Nonbonding (n) orbitals contain unshared electron pairs.
In most of the molecules, the lowest energy occupied molecular orbitals are σ orbitals. The π molecular orbitals have much higher potential energy than σ orbitals in most cases.
σ < π < n < π* < σ*
It can be seen above; how the potential energy of the molecular orbitals changes with respect to one another. But in some special cases like N2 this pattern changes (π < σ < π* < σ*). (Because of the magnetic properties of the molecule – N2 is a diamagnetic molecule)
In molecules, there are few possible electronic transitions. They are explained in the below figures.
| Transition type | λ range (nm) | Examples |
| σ to σ* | < 180 | Alkanes – (CH4 – λmax = 125nm) |
| n to σ* | 150 - 250 | H2O - λmax = 167nm |
| π to π* | 200 - 700 | Unsaturated functional groups |
| n to π* | 200 - 700 | Unsaturated functional groups |
In, organic molecules like alkenes (ex: ethylene, 1,3-butadiene) only transition that occurs within UV – visible range is the π to π* transition. That π to π* transition can be described using an π orbital system diagram. π orbital system of 1,3-butadiene is given below.
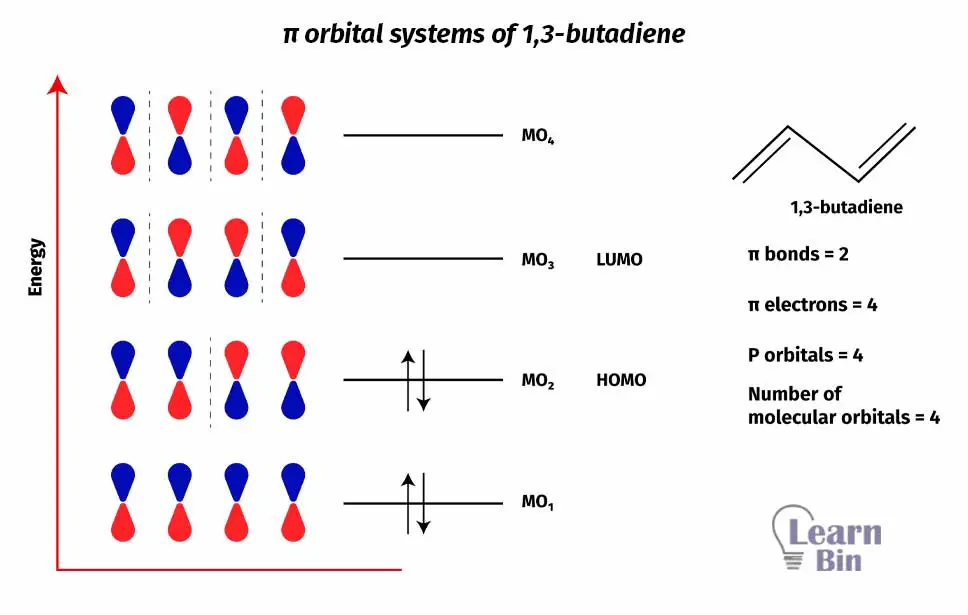
In the above π orbital system diagram, there are 4 molecular orbitals. In this conjugated system, 4 atomic p orbitals that contribute to the π bonds are linearly overlapped to make those molecular orbitals.
When the number of nodes increases, that increases the energy of that molecular orbital. The meaning of the Node is, in the location of a node, there is zero possibility for an electron to exist.
When radiation was absorbed, electrons excite and the transition happens to the LUMO from "HOMO".
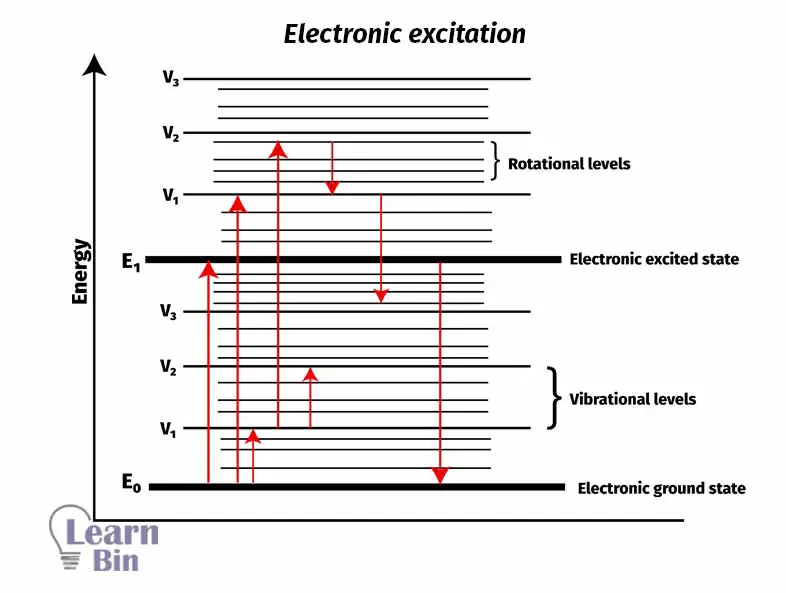
In UV-Vis spectroscopy, when a molecule absorbs energy from light, it excites its electrons to an excited electronic level from the ground state. Molecules absorb a range of wavelengths (range of energy) because molecule also contains vibrational and rotational energy levels.

Alkanes are saturated organic molecules. They don’t have any double bonds or triple bonds or any kind of unsaturation. Also, they don’t contain any unshared electrons. So, the only possible electronic transition that happens in alkanes is σ to σ* transition. As discussed previously, that kind of transition requires much higher energy. They absorb UV radiation at very low wavelengths that cannot even experimentally accessibly using typical spectrophotometers.
All these molecules are saturated molecules that contain non-bonding electron pairs. Because functional groups are saturated, and have non-bonding electrons, the possible electronic transition is n to σ* transitions. They are also high energy transitions (low wavelengths) but they are within the experimentally accessible range of spectrophotometers.
Alkenes (-C=C-) and alkynes (-C≡C-) are unsaturated organic molecules that contain π bonds. So π to π* transitions become possible in these molecules. Alkene absorbs around 175 nm and alkynes absorb around 170 nm.
Also, their λmax and absorption intensity are sensitive to substitutions. (Auxochrome can increase wavelength and absorption)
Carbonyl compound contains both double bond (unsaturation) and non-bonding electron pairs (unshared electrons). So, they can show n to π* transitions and π to π* transitions.
Carbonyl compounds undergo n to π* transitions at 280 to 290 nm range but they are low-intensity transitions because they are forbidden transitions.
Also, carbonyl compounds undergo π to π* transitions at around the 188 nm range.
Also, λmax and absorption intensity of carbonyl compounds are sensitive to substitutions. (Auxochrome can increase wavelength and absorption)
Transitions that have absorption maxima value (εmax) higher than 104 are called allowed transitions. Allowed transitions are generally caused by π to π* transitions.
Transitions that have absorption maxima value (εmax) less than 104 are called forbidden transitions. Forbidden transitions are generally caused by n to π* transitions.
These bands originated from π to π* transitions in the molecules that have a conjugated system. (-C=C-C=C-)
These bands originated from n to π* transitions of a single chromophore of a molecule like a carbonyl or a nitro group.
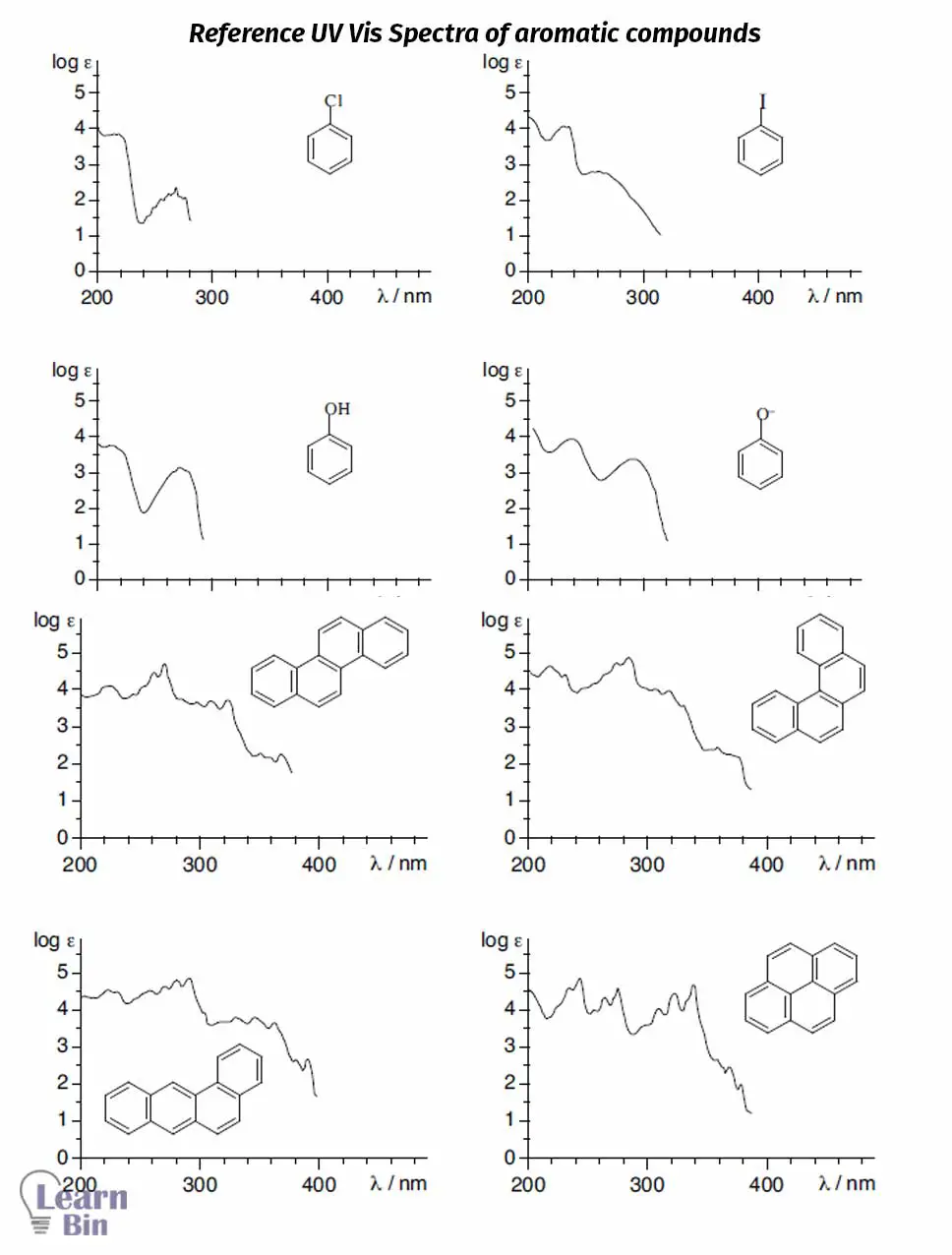
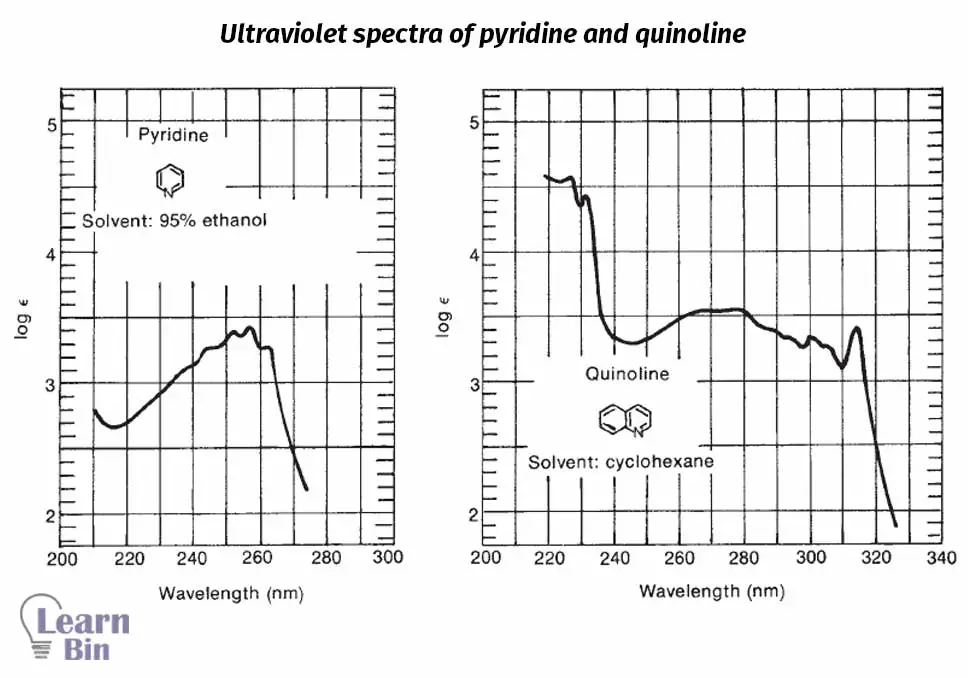
These bands originated from π to π* transitions happen in the aromatic or heteroaromatic molecules.
These bands originated from π to π* transitions happen in ethylenic bonds of aromatic or heteroaromatic molecules.
A = log10 (I0/IT)
A = Absorbance
I0 = Light passing through reference cell (Intensity of the incident beam)
IT = Light passing through sample cell (Intensity of the transmitted beam)
A = 0 means the sample (the compound that is tested) did not absorb any radiation.
A = 1 means the sample has absorbed 90% of the light passing through it.
Beer-Lambert law can be used to calculate the molar absorptivity.
A = ε × C × l
ε = A / (C × l)
A = Absorbance
ε = Molar absorptivity – extinction coefficient (dm3 mol-1 cm-1)
C = Concentration of the sample solution (mol dm-3)
l = Path length (length of the sample cell) (cm)
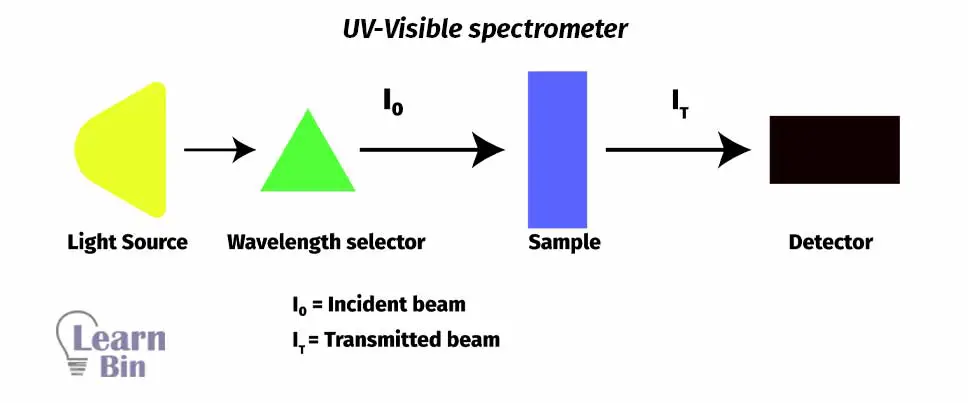
Generally, a UV Vis spectrometer consists of three main parts.
In instruments like double-beam spectrometers, the light beam that exits from the monochromator is split into two beams. One beam is called a reference beam and the other is called a sample beam. The sample beam passes through the cell that contains the actual sample that needed to be analyzed. Reference beam reference cell that only contains the solvent that was used to dissolve the sample.
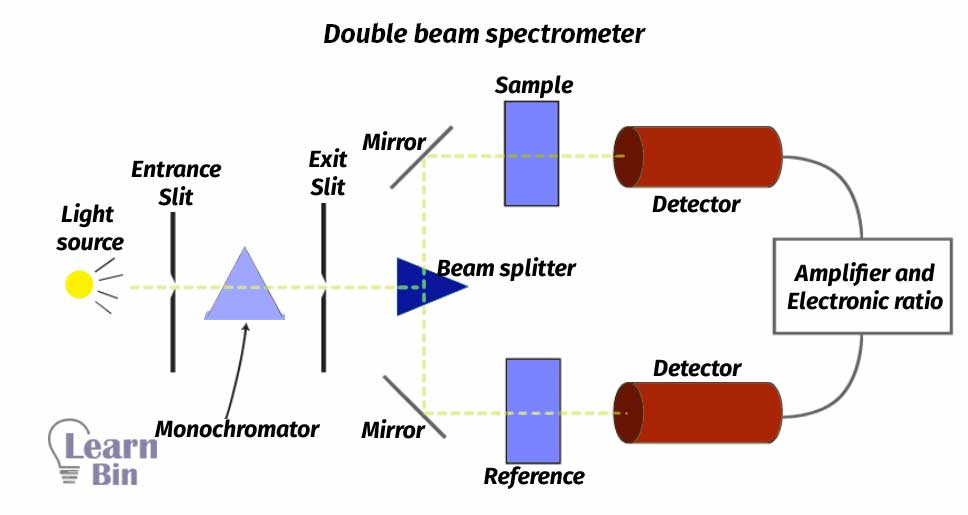
The sample cell must be made out of material that is transparent to UV and visible regions of electromagnetic spectra. The sample cell shouldn’t be absorbing any of the light that passes through the cell. Normally transparent, clear plastic or glass cells can be used to analysis of samples using the visible range of the spectra. But those types of cells cannot use for analysis in UV regions of spectra because those types of cells absorb UV radiation. So, quartz cells are used to analyze samples using UV radiation.
The above-described instrument is only can use to analyze using only one wavelength at a time. If a complete spectrum is required (using a range of wavelengths), this kind of instrument has some difficulties because the monochromator must be mechanically rotated each time that wavelength needs to be scanned. So, these instruments are considerably slow, and a long time is required to acquire the complete spectrum.
But in modern instruments, like “Diode-array spectrophotometer” these kinds of problems have been addressed. This instrument has an array of photodiode detectors that each can scan a narrow band of transmitted spectrum. This kind of detector can record the entire spectrum at once. Also, this kind of instrument has no moving parts. So, this is a more efficient and reliable instrument. Also, the results of the analysis conducted on this machine can be directly passed to a computer and that is helpful in getting more detailed output.

Generally, this kind of spectrum is plotted and recorded using absorbance versus wavelength. The data was replot using molar absorptivity (ε) or log ε on the y-axis and wavelength on the x-axis.
When analyzing a UV-Vis spectrum, clear peak absorbance or ε can be observed. The wavelength that is responsible for the top of that peak is called (wavelength of maximum absorbance) “λmax”.
That wavelength is the wavelength that is responsible for the maximum UV or visible light radiation absorbance of a molecule (its functional groups and conjugations). Molecules use energy from that specific wavelength to excite their electrons to "HOMO" to LUMO. If that was considered as the only transaction, the absorbance value to other wavelengths must be zero. So, it should be one intense sudden peak.
But when looking at UV-Vis spectra, it can be seen some absorbance at most of the wavelengths. That happens because molecules also have vibrational and rotational energy levels. Using energy provided, electrons can transmit to those levels as well.
UV or visible light absorption of a molecule is happening because electrons are excited to a higher energy level from the ground state. But the nuclei of those atoms are the deciding factor of which wavelength should be absorbed. The reason for that is nuclei (charge of the nuclei) is the factor that decides how strong electrons should interact with the atom and so it will decide the energy gap between the energy levels.
So, the energy gap between energy levels and hence the wavelength of radiation that is absorbed by a molecule is a property of a group of atoms rather than electrons electron themselves. The Group of atoms that are responsible for that kind of absorption (responsible for absorption of light/ radiation) is called a “Chromophore”. Generally, the above-mentioned group of atoms that are responsible for UV or visible light absorption is a covalently unsaturated group of atoms. (-C=C-, -C≡C-, -C=O, -C≡N, N=N, NO2)
In some references, chromophores are described as the part of the molecule that is responsible for its color. But this is a partially correct statement because if the molecule (chromophore of that molecule) absorbs radiation in the visible range (400 – 800nm), only then molecule/ compound appears as colored. So, the chromophore of the molecule may or may not impact its color depending on its absorption of UV or visible light.
It is very difficult to determine how absorption will change the structure of chromophores and their behavior. So, to predict that kind of relationship, it has to be dependent on empirical (observational and experimental) working guides.
| Chromophore | Example | λmax (nm) | εmax (dm3 mol-1 cm-1) | Transition | Solvent |
| -C=C- | Ethylene | 171 | 15 530 | π → π* | Vapor |
| -C≡C- | Acetylene | 150 | 10 000 | π → π* | Hexane |
| 173 | 6000 | π → π* | Vapor | ||
| -C=O | Acetaldehyde | 160 | 20 000 | n → σ* | Vapor |
| 180 | 10 000 | π → π* | Vapor | ||
| 290 | 17 | n → π* | Hexane | ||
| Acetone | 166 | 16 000 | n → σ* | Vapor | |
| 188 | 900 | π → π* | Hexane | ||
| 279 | 15 | n → π* | Hexane | ||
| -COOH | Acetic acid | 204 | 60 | n → π* | Water |
| -CONH2 | Acetamide | 178 | 9 500 | π → π* | Hexane |
| 220 | 63 | n → π* | Water | ||
| -COOR | Ethyl acetate | 211 | 57 | n → π* | Ethanol |
| -NO2 | Nitromethane | 201 | 5 000 | π → π* | Methanol |
| 274 | 17 | n → π* | Methanol | ||
| -C=N- | Acetoxime | 190 | 5 000 | n → π* | Water |
| -C≡N | Acetonitrile | 167 | weak | π → π* | Vapor |
| -N=N- | Azomethane | 338 | 4 | n → π* | Ethanol |
Some chromophores of organic compounds contain covalently saturated substituent groups replacing their hydrogen atoms. Those attachments of chromophores change the wavelength and intensity of the absorbance of absorption maxima of chromophores. That kind of attached group is called “auxochrome”.
Generally, those auxochrome increases absorption maxima and λmax of chromophores by extending conjugations by resonance.
There are four kinds of effects/ shifts on the absorption spectrum that can be observed because of substituents and conjugations of the molecule.
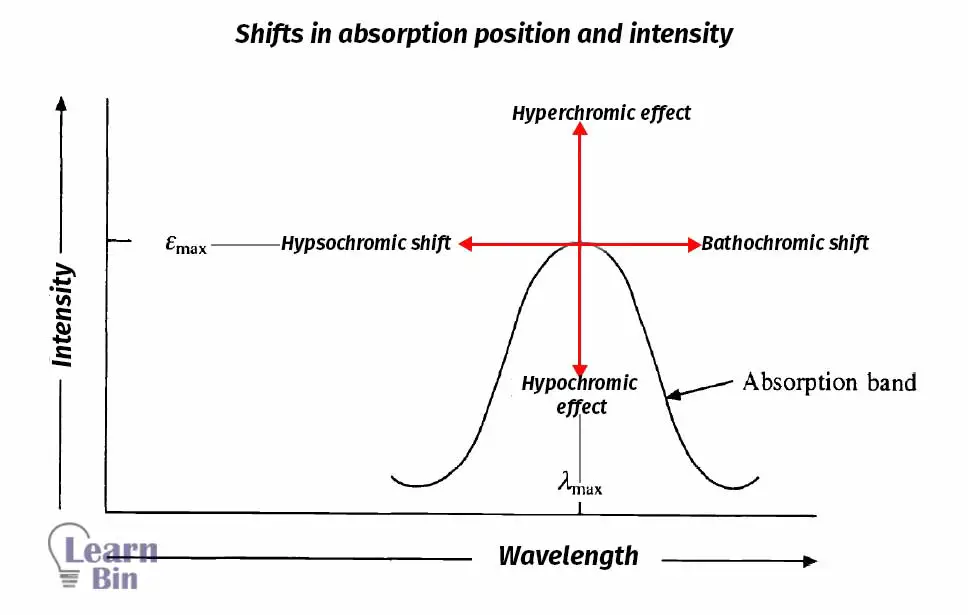
The shift of the absorption maxima to a longer wavelength (lower energy) is called a bathochromic shift.
This shift can be occurred due to the presence of an auxochrome, increasing conjugations, or solvent effect.
The shift of the absorption maxima to a shorter wavelength (higher energy) is called a hypsochromic shift.
This shift can be occurred due to the removal of conjugation or a change in the solvent polarity.
An increase in absorption intensity is called a hyperchromic effect.
This effect can be occurred due to the introduction of an auxochrome.
A decrease in absorption intensity is called a hypochromic effect.
This effect can be occurred due to the introduction of a group that has the ability to distort the chromophore.
Conjugation means that there are linked π systems present in that molecule. The meaning of that is there are adjacent double bonds. Using the below figure, this can be understood.
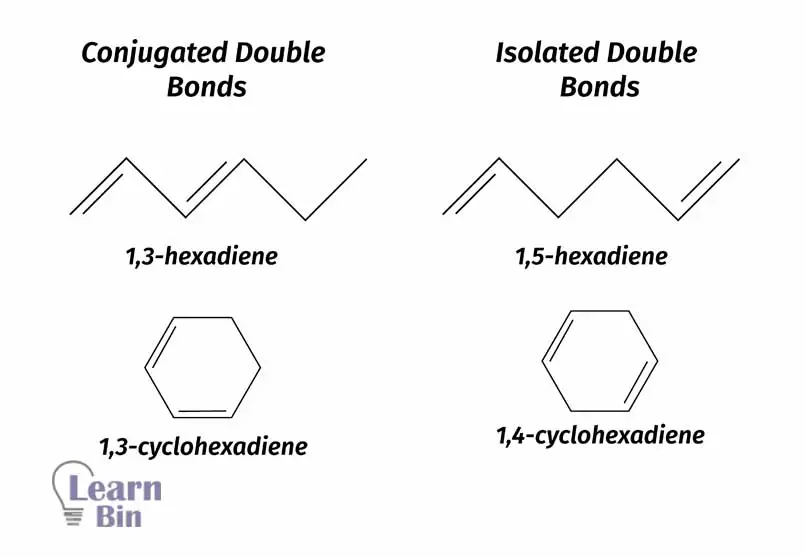
When conjugation increases, the energy gap between "HOMO" and LOMO decreases. That means λmax increases when conjugation increases because energy is inversely proportional to the wavelength. That means the presence of a conjugated system increases the bathochromic shift.
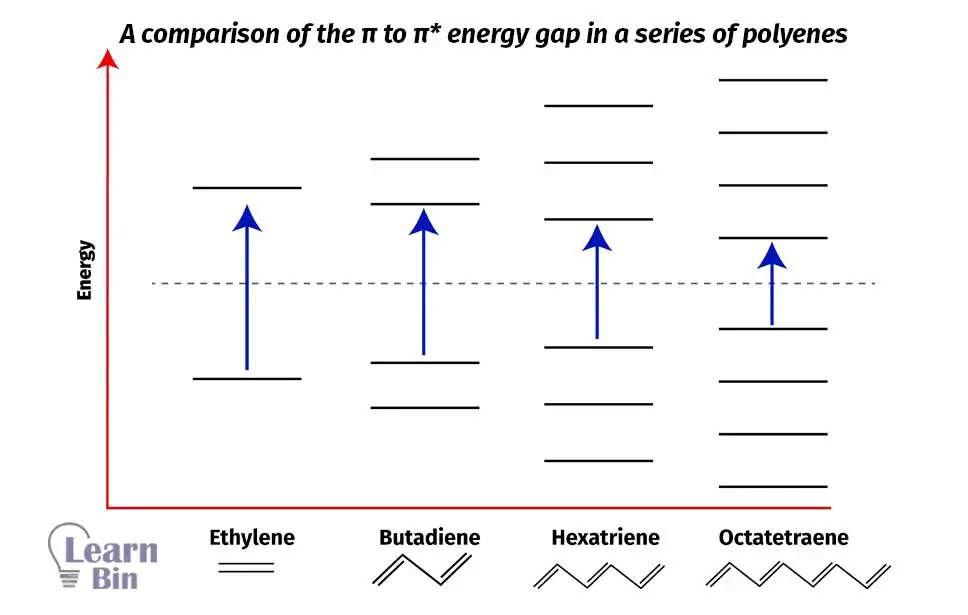
When conjugated chromophores are present that not only results in a bathochromic shift but also increases the intensity of absorption.
| Molecule | λmax (nm) | εmax (dm3 mol-1 cm-1) |
| Ethylene | 175 | 15 000 |
| 1,3-butadiene | 217 | 21 000 |
| 1,3,5-hexatriene | 258 | 35 000 |
Choosing the right solvent for UV-vis spectroscopy is very important. Solvents are used to dissolve samples that UV vis spectrum is required. Solvent to use in UV vis spectroscopic analysis, there are a few requirements/criteria to be considered.
01. Solvent should not absorb the UV radiation in the same range of compound that spectrum is determined. So, solvents that don’t contain any conjugated systems are much more suitable.
02. It should be determined that how solvent affects the fine structure of the absorption band. There are two kinds of solvents. One is polar solvents and the other is nonpolar solvents.
Nonpolar solvents do not form hydrogen bonds with the compound (solute). So, it would not affect the fine structure of the absorption band. So fine absorption band can be observed.
But polar solvents form hydrogen bonds with the compound and will form solvent-solute complexes. Then, that would affect the fine structure of the absorption band of the complex (molecule).
Polar solvent shifts π to π* transitions of polar compounds towards higher wavelength and higher absorption intensity. Also increasing the polarity of a solvent would result in shifting n to π* transitions to a shorter wavelength.
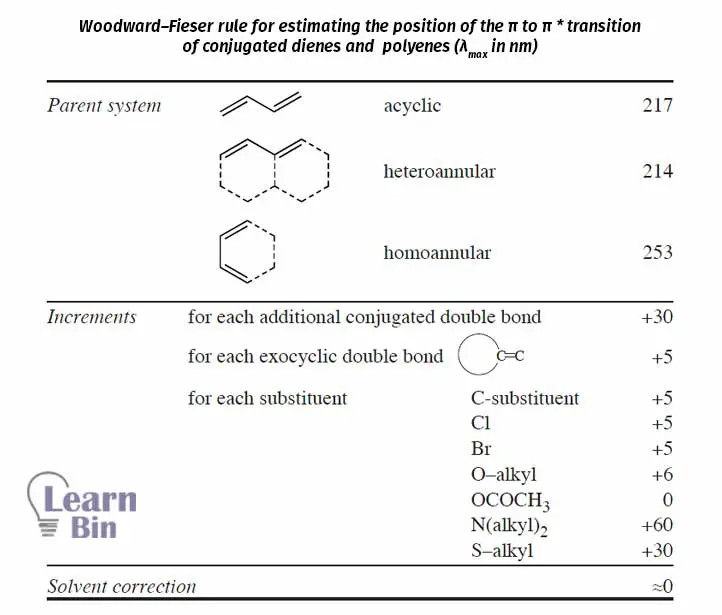
The Woodward-Fieser rules are several sets of rules that use to predict λmax (Wavelength at the absorption maxima) of a UV visible spectrum of a specific molecule/ compound.
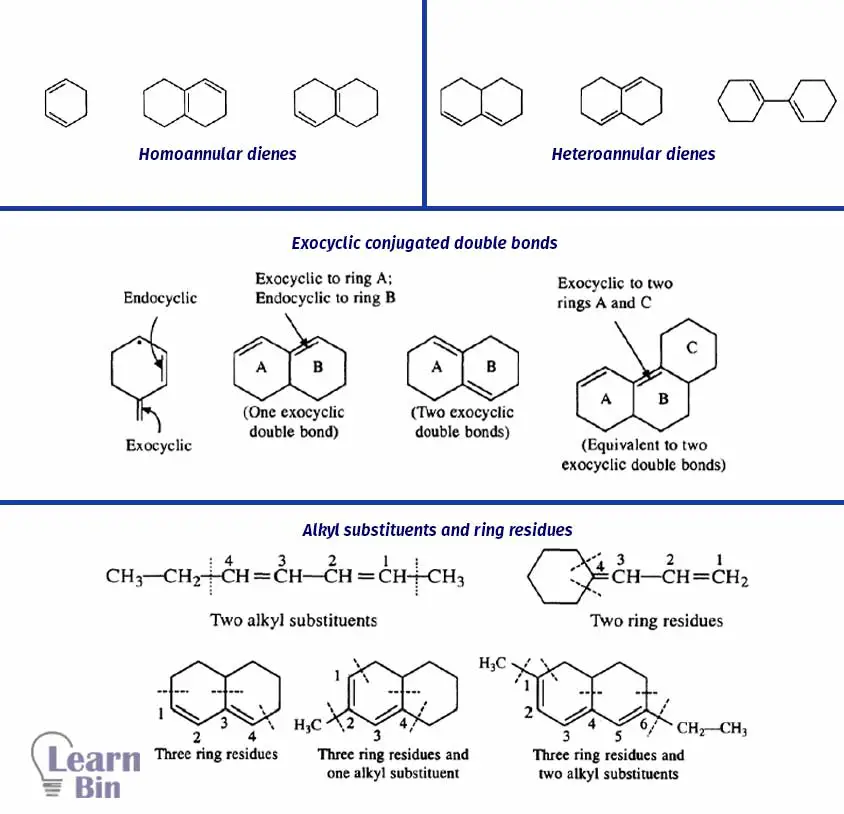
Before describing the calculations, some specific terminologies are important to know.
1. Homoannular dienes
Conjugated double bonds should be present in the same ring. They have s-cis confirmation.
2. Heteroannular dienes
Conjugated double bonds are not present in the same ring. They have s-trans conformation.
3. Exocyclic conjugated double bonds
Carbon-carbon double bond should project outside of the ring
4. Alkyl substituents and ring residues (C – Substituents)
Only ring residues and alkyl substitutions that are attached to the carbon atoms of the conjugated system are considered
Using this data, the position (λmax) of π → π* transition of dienes and polyenes can be estimated using Woodward–Fieser rules.
Example:
01. Estimating λmax of Myrcene
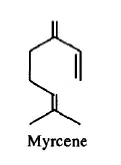
Base value (Acyclic molecule) – 217 nm
1 C – Substituent - 5 nm
Estimated value = 222 nm
Experimental value = 225 nm
02. Estimating λmax of 1,2,3,5-Tetrahydro-5-isopropyl naphthalene
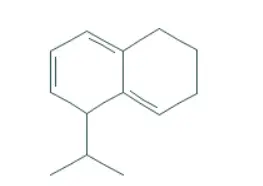
Base value (homoannular molecule) – 253 nm
1 additional conjugated double bond – 30 nm
2 exocyclic double bonds – 2*5 nm – 10nm
4 C- substituents (ring residues) – 4* 5nm – 20nm
Estimated value = 313 nm
Using this data, the position (λmax) of π → π* transition of α, β – unsaturated carbonyl compounds can be estimated using Woodward–Fieser rules.
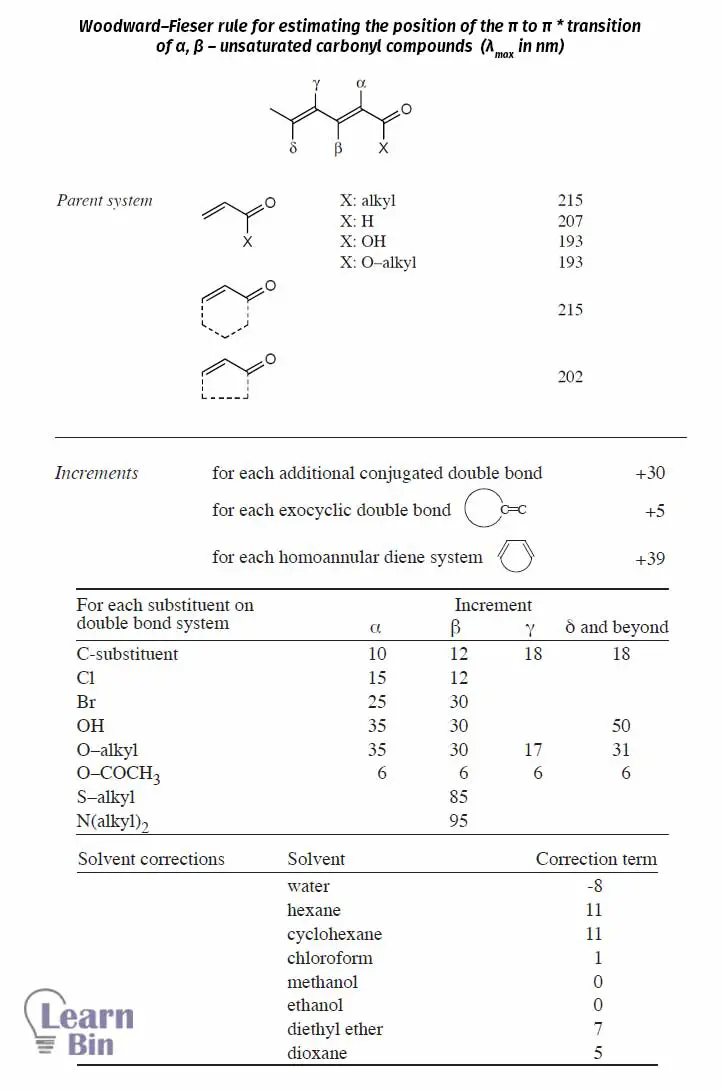
Example:
01. Estimating λmax 1-acetyl-1-cyclohexene (in methanol)
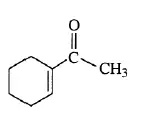
Base value ( α, β – unsaturated ketone) = 215 nm
1 C-substituent at α (ring residue) = 10 nm
1 C-substituent at β (ring residue) = 12 nm
Solvant correction = 0
Estimated value = 237 nm
Experimental value = 232 nm
Using this data, the position (λmax) of the “k band” of the aromatic carbonyl compounds in ethanol solvent can be estimated.
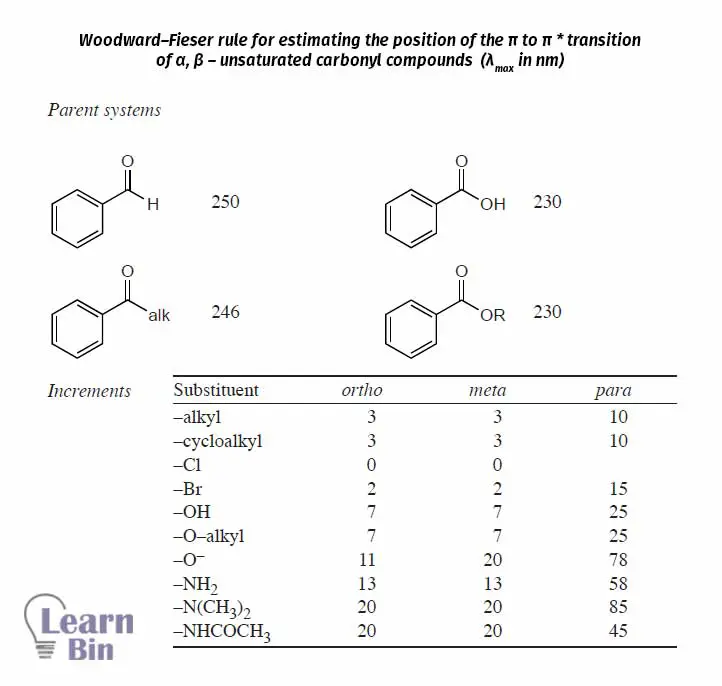
In the electromagnetic spectrum, a wavelength range between about 400 nm to 750nm is called visible light. Humans' eye is sensitive to this range of wavelengths and can detect colors. The light that is between 400 nm wavelength region appears as violet color and light around 750nm appears as red color.
When considering the colors of the compounds, if the compound absorbs visible light, that compound will have a color. If the compound did not absorb visible light, it won’t show any color and will appear as white.
But in the compounds that absorb visible light, the color that absorbs by the compound is not the color that the compound possesses. That is because, if we shine light in visible range into a substance, that substance absorbs a certain wavelength or range of wavelengths of radiation, and the rest of the wavelengths will be reflected. So, we only can observe the reflected wavelengths and their color only. Color that is absorbed by the substance is not observable. In a transparent substance, we can only detect transmitted wavelengths from that substance.
Compounds that contain highly extended conjugated electron systems show this kind of relationship with visible light and they can have colors. Highly extended conjugated systems of those compounds have shifted their absorbance wavelengths into much higher value and into the visible range and because of that, they can appear as colored.
| Absorbed color | The wavelength of the color absorbed (nm) | Observed color |
| Violet | 400 | Yellow-green |
| Indigo blue | 425 | Yellow |
| Blue | 450 | Orange |
| Blue-green | 490 | Red |
| Green | 510 | Purple |
| Yellow-green | 530 | Violet |
| Yellow | 550 | Indigo blue |
| Orange | 590 | Blue |
| Red | 640 | Blue-green |
| Purple | 730 | Green |
Examples:
β-carotene (pigment of carrot)
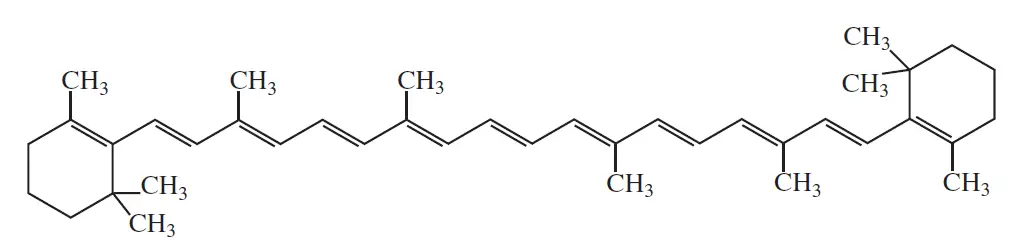
λmax =452nm | Color = orange
Cyanidin (pigment of cornflower)
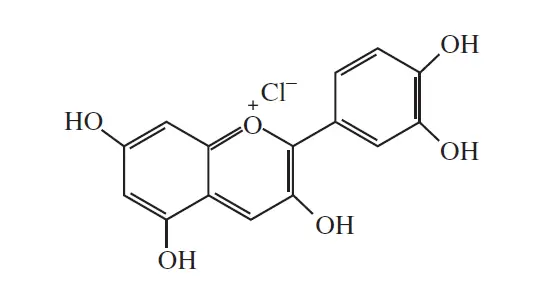
λmax =545nm | Color = blue
Malachite green
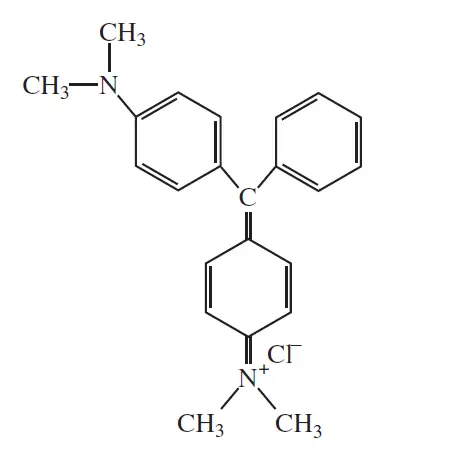
λmax =617nm | Color = green
The ultraviolet spectrum doesn’t provide many details about the structure of a substance that is analyzed in UV-vis spectroscopy. The Ultraviolet spectrum is useful when you have at least a general idea about the compound that is tested.
If you combined results from the Ultraviolet spectrum with some other advanced spectroscopic method like NMR or infrared, that would become very useful. For example, then those data can be used for purposes like identifying carbonyl groups, double bonds, aromatic rings, and other important chromophores.
Following observations can be used as guidelines when it comes to analyzing the Ultraviolet spectrum.

[1]Tables of Spectral Data for Structure Determination of Organic Compounds. 4th ed. - Pretsch, E., Biemann, K., Clerc, T., Seibl, J., Simon, W. and Pretsch, E., 2009.
[2]Introduction to spectroscopy. 5th ed - Pavia, D., Lampman, G., Kriz, G., Vyvyan, J. and Lin, T., 2015.
Myrcene - National Center for Biotechnology Information (2021). PubChem Compound Summary for CID 31253
1,2,3,5-Tetrahydro-5-isopropylnaphthalene - National Center for Biotechnology Information (2021). PubChem Compound Summary for CID 101316547
The cover image was designed by using an image from Matylda Sękpl.wiki: Cygaretkacommons: Cygaretka, CC BY-SA 3.0, via Wikimedia Commons
Figure 08: Image by Viv Rolfe, CC BY-SA 4.0, via Wikimedia Commons
Figure 18: Image of the spectra is based on data in Ultraviolet Spectra of Aromatic Compounds, John Wiley and Sons, New York, 1951
How the double bond belongs to first ring if it is been shown here in the second ring ? If you show the double bond in the second ring it means observer assumes it belongs to the second ring , then it can't be homoannular. If you show the double bond in first ring then the observer assumes it is in first ring then it can be homoannular.Please explain it.
It is a common thing in chemistry to show structures like this. If you refer chemistry text books you can see this kind of representations commonly. It is not wrong to do so.
Just take a piece of paper and draw to carbon atoms and bonds of the ring structure. You can see the structure and where the double bonds are and where they belong. It will a good exercise to understand the ring structures of hydrocarbons.
How the double bond belongs to first ring if it is been shown here in the second ring ? If you show the double bond in the second ring it means observer assumes it belongs to the second ring , then it can't be homoannular. If you show the double bond in first ring then the observer assumes it is in first ring then it can be homoannular.
In Chromophore and their properties table 02 , the acetic acid has also the possibility of doing pi to pi*, sigma to pi* rather than only
n to pi* .So why the other two have not been mentioned In the table where for the aldehyde and acetone you have mentioned all three possibility.
Thank you for your comment and your question is a good one. The reason for not observing π to π* transition in carboxylic acids (in most carbonyl compounds also) is due to the effect of the solvent. Most of the time only n to π* transitions are visible in the UV-visible spectrum, above the solvent cutoff point. So in the UV spectrums of most carboxylic acids and carbonyl compounds, only n to π* transitions are taken into consideration. If you look at the UV-vis spectrum of acetic acid, you can see this. There are only weak n to π* transitions present in there. This doesn't mean π to π* or other transitions won't occur. Only they won't occur within the operational range of UV-visible spectrometer.
I hope this answer will clear things up for you. If you have any more questions Please let us know.
Thanks for your acetic acid answer. Just a suggestion, please mention it in somewhere around the table 02 it would be helpful.
Another question Figure 12: Terminologies related to dienes, In homoannular dienes , the third homoannular ring , how it can be homoannular because homoannular dienes definition says that both the double bonds must be in the same ring , it must be a heteroannular ring.
Thank you for your suggestion and in the future we will add more articles related to these topics to make more in-depth explanations.
About the homoannular diene that you are referring to, if you take a look carefully, the second double bond belongs to both rings. And also it shows cis conformation. So in conclusion, both double bonds are present in the same ring. That is why it is a homoannular diene. To become a hetero-annular diene "Both double bonds should not present in the same ring". I hope this will clear things up for you.