More results...


In analytical chemistry, chemicals are analyzed qualitatively or quantitatively. Simply, qualitative analysis is checking whether there is a chemical compound present or not. Quantitative analysis is the determination of how much that compound is present in the sample.
To identify cations that are dissolved in water, a precipitation method can be used. In this method, cations are classified into five groups.
| Group | Cations |
| 1 | Ag+, Pb2+, Hg22+ |
| 2 | As3+, Sb3+, Bi3+, Sn2+, Sn4+, Hg2+, Pb2+, Cu2+, Cd2+ |
| 3 | Cr3+, Al3+, Fe3+ |
| 4 | Zn2+, Mn2+, Ni2+, Co2+ |
| 5 | Ca2+, Sr2+, Ba2+ |
Group 1 cations are identified by precipitating as their chlorides. When an aqueous solution of cations is added to diluted HCl acid, a white color precipitate is formed if group 1 cations are present.
In the analysis of group 1 cations, concentrated HCl cannot be used. Because PbCl2 will dissolve in concentrated HCl and form an ion complex as follows.
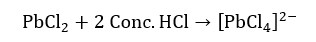
PbCl2 also dissolves in hot water. Therefore, hot water cannot be used in the analysis of group 1 cations. If a cations solution is added diluted HCl and there is no precipitate formed, it concludes that there are no group 1 cations in that solution.
If there is a precipitate formed in the group 1 analysis, the filtrate is taken for the analysis process for group 2 cations. If there is no precipitate from the group 1 analysis, the solution is directly taken for the group 2 analysis.
The filtrate of group 1 analysis is bubbled Hydrogen sulfide (H2S) gas followed by diluted HCl acid. The reason for adding HCl is to reduce the concentration of sulfide ions. The H+ ions released from HCl shift the equilibrium of H2S backward. Thus, the concentration of the sulfide ion is decreasing.
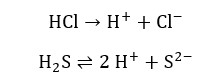
Group 2 cations are precipitated as the respective sulfides. These sulfides have relatively low Ksp values. Therefore, they will precipitate at low concentrations of sulfide ions. (S2-).
| Precipitate | Color of the precipitate |
| PbS | Black |
| CuS | Black |
| HgS | Black |
| Bi2S3 | Black |
| SnS | Brown |
| SnS2 | Yellow |
| CdS | Yellow |
| As2S3 | Yellow |
| Sb2S3 | Orange |
The filtrate of the group 2 analysis is taken for the group 3 analysis. If there is a precipitate formed in group 2, it is separated first. In group 3 there are three cations which are Cr3+, Al3+, and Fe3+. These cations will be precipitated as their respective hydroxides at low concentrations of hydroxide ions (OH-)
| Precipitate | Color of the precipitate |
| Cr(OH)3 | Green |
| Al(OH)3 | White color cloudy |
| Fe(OH)3 | Reddish brown |
Qualitative analysis of group 3 cations is done in 3 steps.
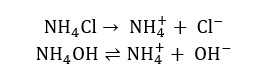
Due to the NH4+ ions coming from NH4Cl, the dissociation of the NH4OH is reduced. Therefore, the concentration of the hydroxide ions in the solution is reduced.
Thus, the cations that are precipitated at low concentrations of OH- ions will be precipitated in group 3 analysis. If the concentration of OH- ions is high, these precipitates will dissolve in the solution.
If there is any precipitate formed in group 3 analysis, it is separated and the filtrate is taken for the group 4 analysis. There are four cations in group 4 which are Zn2+, Mn2+, Ni2+, and Co2+. These cations will precipitate as their respective sulfides at high concentrations of sulfide ions (S2-).
| Precipitate | Color of the precipitate |
| ZnS | White |
| MnS | Pink |
| NiS | Black |
| CoS | Black |
The solution is added Ammonium hydroxide solution (NH4OH) and bubbled with Hydrogen sulfide (H2S) gas. The purpose of adding NH4OH is to increase the concentration of sulfide ions (S2-) ions in the solution.
According to Le Chatelier’s principle, the equilibrium will shift to the right and it will result in a high concentration of S2- ions in the solution.
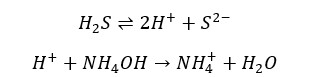
These precipitates will dissolve in dilute acids.
If there is any precipitate formed in the group 4 analysis, it is separated and the filtrate is taken for the group 5 analysis. There are three cations in group 5, which are Ca2+, Sr2+, and Ba2+. These cations will be precipitated as their respective carbonates.
| Precipitate | Color of the precipitate |
| CaCO3 | White |
| SrCO3 | White |
| BaCO3 | White |
The qualitative analysis of Group 5 is done in three steps.
When considering the filtrate of the group 5 qualitative analysis, it may present any other cations except those cations from groups 1 – 5.

The cover image was created using an image by RF._.studio from Pexels