More results...


Separation is a process by which the components are separated from their mixture by using different techniques. Amino acids occur in D and L forms except for glycine. L – amino acids are biologically used by cells while pure D-amino acids are industrially important and used in the synthesis of drug intermediates, pharmaceuticals, and food ingredients.
When amino acids are synthesized artificially, a mixture of L and D enantiomers is formed. Sometimes properties of amino acids depend on the 3D orientation of the molecule. In most cases, only the L enantiomers are biologically active, and the D enantiomers may even be toxic. So, it is important to separate amino acids before using them. And separation of the pure form of amino acids (D- or L-form) is the most challenging task.
Pure L enantiomers are needed for peptide synthesis for the activity of the natural material. Because both L and D enantiomers have the same physical properties, the separation is much more difficult.
To separate amino acids, chromatography is mostly used. Two general chromatographic methods for the separation of amino acids by GC are
Gas chromatography is a type of partition chromatography. The mobile phase is a chemically inert and non-reactive gas like Helium, Argon, Nitrogen, etc. the choice of the mobile phase (carrier gas) is dependent on the type of detector used for the detection of the separated analyte. The stationary phase of gas chromatography is a liquid.
First, the analyte interacts with the stationary phase. Depending on the interaction, molecules of the analyte will retain the stationary phase. If the interaction between the analyte and stationary phase is weaker, molecules will quickly move with the mobile phase.
The components that are more soluble in the stationary phase interact strongly with the stationary phase and elute later. The components that are comparatively less soluble will elute faster. The separation of those components depends on the interaction with the partition coefficient between the stationary phase and the analyte.
In this method, the enantiomer mixture is reacted with another chiral molecule. This will result in a diastereomeric mixture (A mixture of compounds that have two chiral centers).
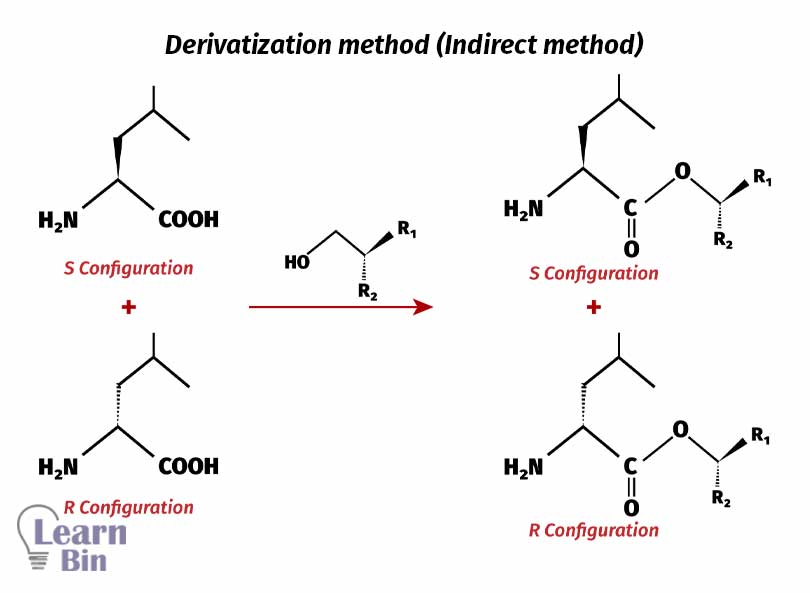
According to the stereochemistry of the molecule that is reacted with the racemic mixture, we will get a diastereomeric mixture with S-R and R-R molecules. Diastereomers have different physical properties. So, we can use these physical properties to separate them. After separation, diastereomers can convert into their original molecules.
Here, no chemical reaction is performed prior to the chromatographic separation. In this method, a chiral stationary phase is used. The separation takes place due to the association of reversible diastereomers between the enantiomers and the chromatographic chiral environment.
Widely using chromatographic techniques for amino acid separation in the industry are,
GC is a very effective and sensitive technique for amino acid separation. The only limitation that occurred with GC is the amino acids that are to be separated must be derivatized to the volatile compound and amino acids required a special condition for derivatization. The derivatization process is a multi-step process. It consists of removal of water; esterification; removal of excess alcohol, then acylation and removal of all the excess reagents.
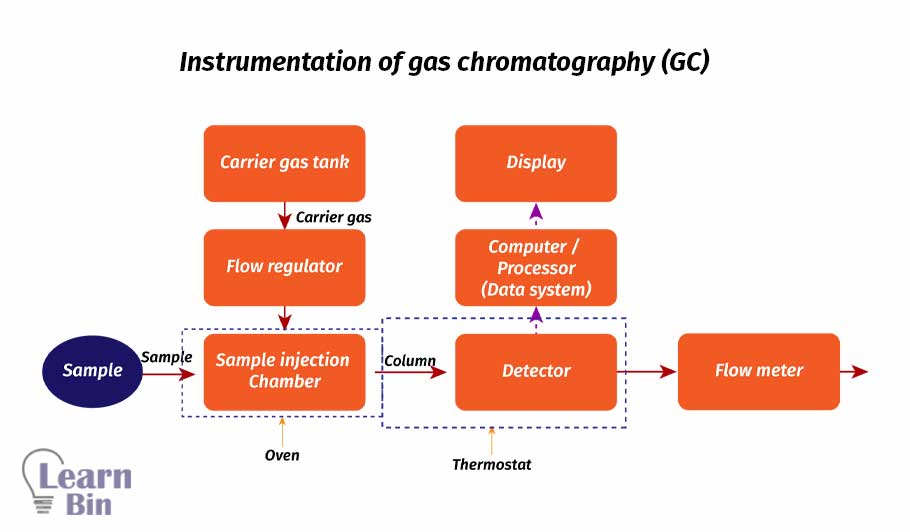
High-performance liquid chromatography or high-pressure
liquid chromatography is the widely used method in industry to separate amino acids. It is an analytical technique used to separate, purify, identify, and quantity of an unknown component from a complex mixture.
There are two types of HPLC depending on the stationary phase,
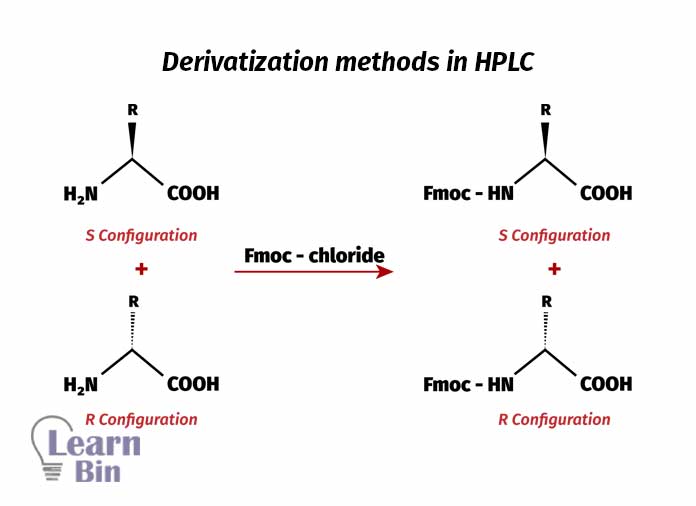
Derivatization methods also can be used in HPLC. Here, the racemic mixture of amino acids is reacted with Fmoc chloride. So, the nitrogen group in amino acid will react with the carbonyl group in Fmoc chloride (9-Fluorenylmethyloxycarbonyl chloride) and give Fmoc group to the amino acid.
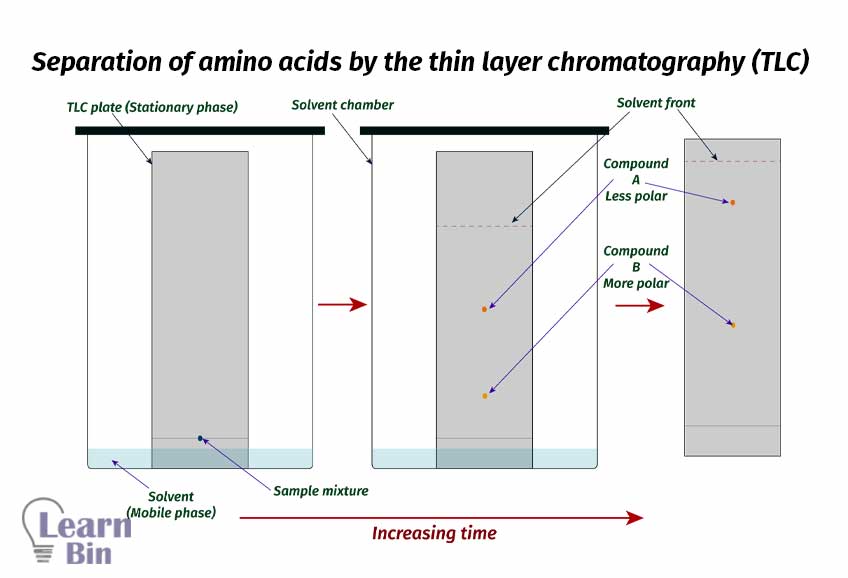
Thin layer chromatography (TLC) is done to separate compounds with different polarities. Enantiomers cannot be separated by the TLC method. Because both enantiomers have the same polarity.
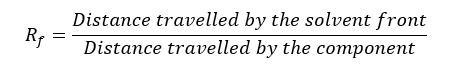
Less polar components have lesser Rf values and high polar components have higher Rf values.
When it comes to the amino acid it may not be separated by TLC. Because amino acids are highly polar molecules. So, sometimes they may not travel through the column. But there are some specific solvents that can be used to separate amino acids.
e.g., acetic acid n-butanol and water system. – 4 parts of acetic acid, 4 parts of n-butanol, and 1 part of water.
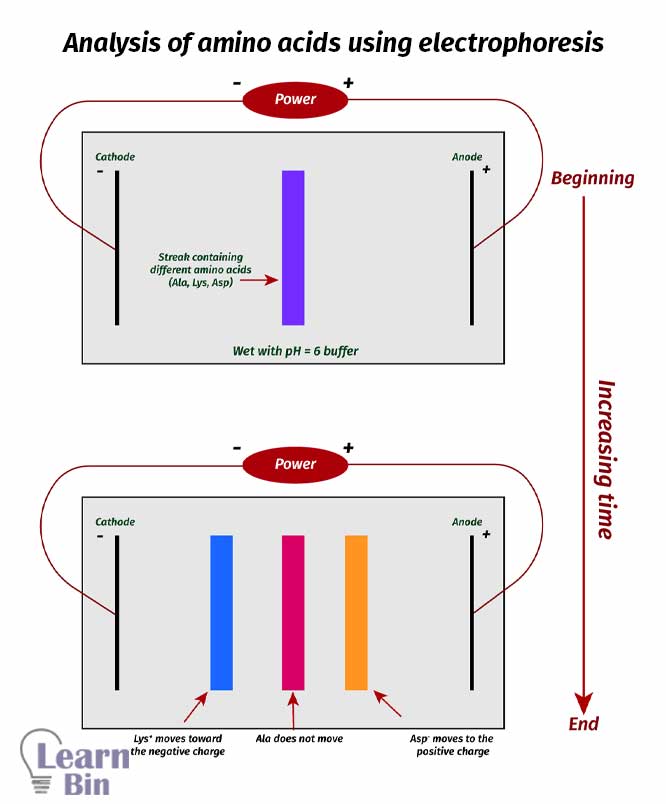
In electrophoresis, differences in isoelectric points are used to separate the mixture of amino acids.
| pH level | Amino acid | PI |
| Acidic | Aspartic acid | 2.8 |
| Glutamic acid | 3.2 | |
| Neutral | Neutral amino acids | 5.0 - 6.3 |
| Basic | Lysine | 9.7 |
| Arginine | 10.8 | |
| Histidine | 7.6 |
A streak of the amino acid mixture is placed on the center of a layer of acrylamide gel, or a piece of filter paper wet with buffer solution. Two edges of the gel or paper are introduced electrodes. Then a voltage of several thousand is applied. So, cationic amino acids will attract to the cathode and anionic amino acids will attract to the anode. Amino acids at their PI have no net charge; so, they don’t move.
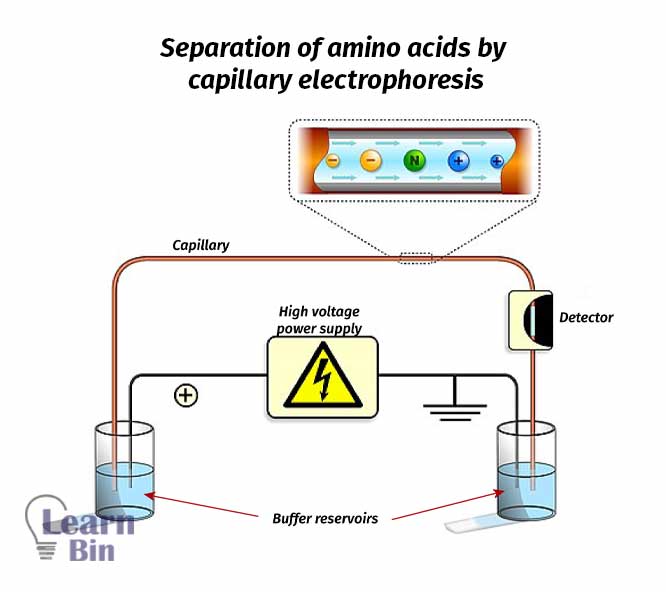
This is the most popular method of amino acid separation. Separation is occurred based on the rate of migration of components under the applied electric field. According to the isoelectric points of amino acids, they will enter the capillary region.
Several electrophoresis methods,
Here, chiral reagents are used in the zone (electrophoresis medium). The separation of enantiomers largely depends on their association with other chiral molecules called the chiral selector. Amino acid molecules will attract to that chiral selector. That interaction also dictates their movements in the electrophoresis medium. Amino acids and chiral selectors will form a complex. This complex migrates at a different rate than the analyte alone.
The enantiomer which has higher interaction with the chiral selector can be separated from the other enantiomer which has a weaker association with the chiral selector.
Hyphenated techniques are a combination of two different techniques with the help of an appropriate interface.
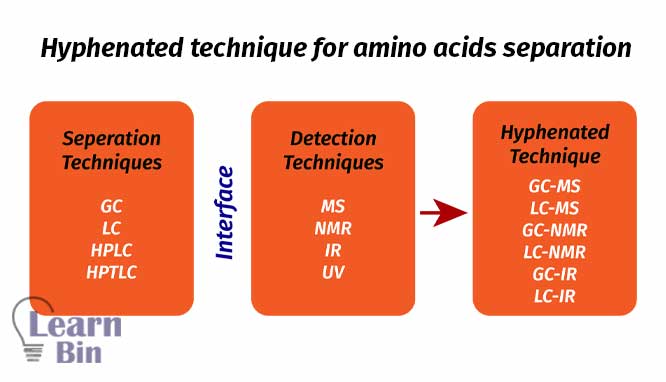
Advantages of hyphenated techniques,
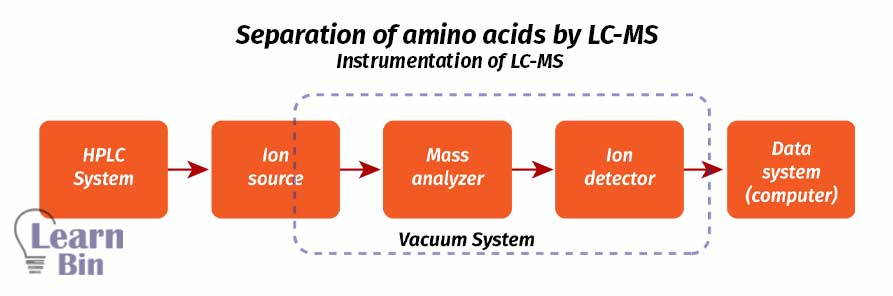
The most popular and powerful hyphenated technique for amino acid separation is LC-MS. It is a combination of Liquid chromatography and Mass spectroscopy. LC separates components from the complex mixture and Mass spectroscopy identifies it. Here, the sample is separated as well as identified. A very small amount of sample is used for analysis in LC-MS.
LC-MS technique is a very sensitive, specific, and selective method. It is suitable for analyzing polar, ionic, thermally unstable, and non-volatile substances.
LC-MS can be applied for components with a lack of chromophores. LC-MS has three major components which are chromatography, interface, and spectrometry. In the HPLC system, components are separated. Then the separated components are transferred to the interface and finally transferred to the mass analyzer. In a mass analyzer, compounds are identified based on their mass.
Separation by CE-MS mainly depends on the electrophoretic mobility and structural information of the amino acids. The combination of capillary electrophoresis and mass spectroscopy stands for CE-MS. CE-MS techniques have been widely used for the separation and identification of the components, owing to their high separation efficiency. This combination of techniques has become very popular in the field of biological sciences and the pharmaceutical industry.
When it comes to the separation of amino acids, based on the pH of the media amino acids will move toward the respective electrode. So, the movement or apparent mobility is given by,
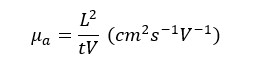
Where,

The cover image was designed using images by Polimerek (licensed under CC BY-SA 4.0 via Wikimedia Commons), Andreas Dahlin (licensed under CC BY 2.0, via Wikimedia Commons), Oak Ridge National Laboratory (licensed under CC BY 2.0), and ConHelius, (licensed under CC BY-SA 2.5, via Wikimedia Commons)
Figure 6: Contains an image by Andreas Dahlin, licensed under CC BY-SA 2.0, via Wikimedia Commons