More results...


Proteins are a large group of biopolymers. The repeating unit also known as the monomer, of proteins is “Amino acids”. Amino acids have connected to each other by “Peptide /Amide bonds” and form peptide chains. When two amino acids are bonded, it is called “Dipeptides”, when three amino acids are bonded, “tripeptides" are formed. When more amino acids are formed in long chains, “Polypeptide chains” are formed.
Proteins are made out of one or more polypeptide chains. Carbon, Hydrogen, Oxygen, and Nitrogen are the key elements found in proteins, and some proteins have sulfur.
Amino acids are the structural units/ repeating units/monomers of proteins. In amino acids, a carbon atom is surrounded by an Amine group (-NH2), a Carboxyl group (-COOH), a hydrocarbon chain (-R), and a Hydrogen atom. This carbon atom is called α carbon. The difference between amino acids is caused by the difference in the hydrocarbon group (R). The simplest amino acid is “Glycine” which has a Hydrogen atom as the “R” group. Sometimes, Carboxyl groups, amine groups, and sulfur atoms are attached to the “R” group.
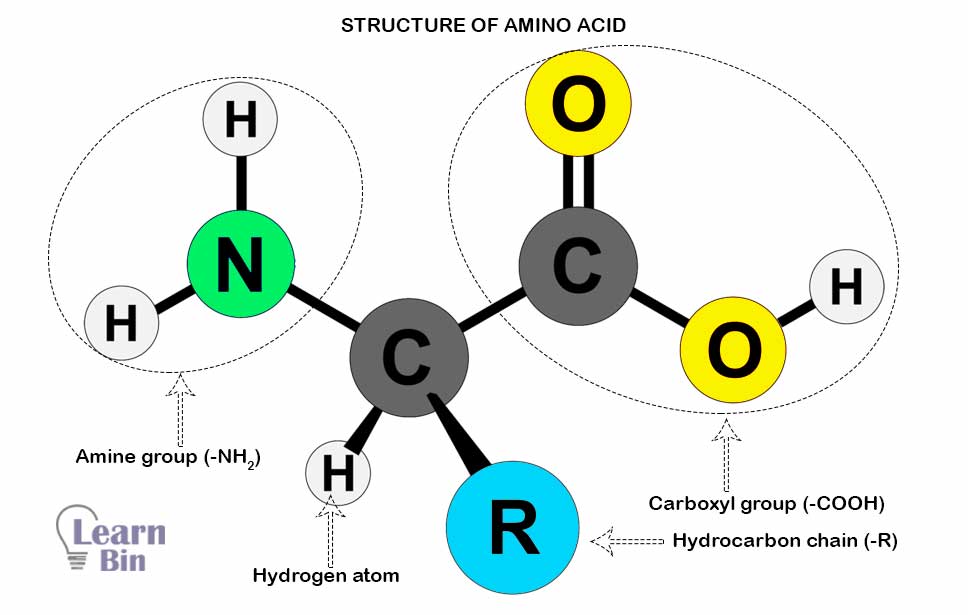
The Carboxyl group has acidic properties, and the amine group has basic properties. Therefore, amino acids have both acidic and basic properties. (Amphoteric compounds)
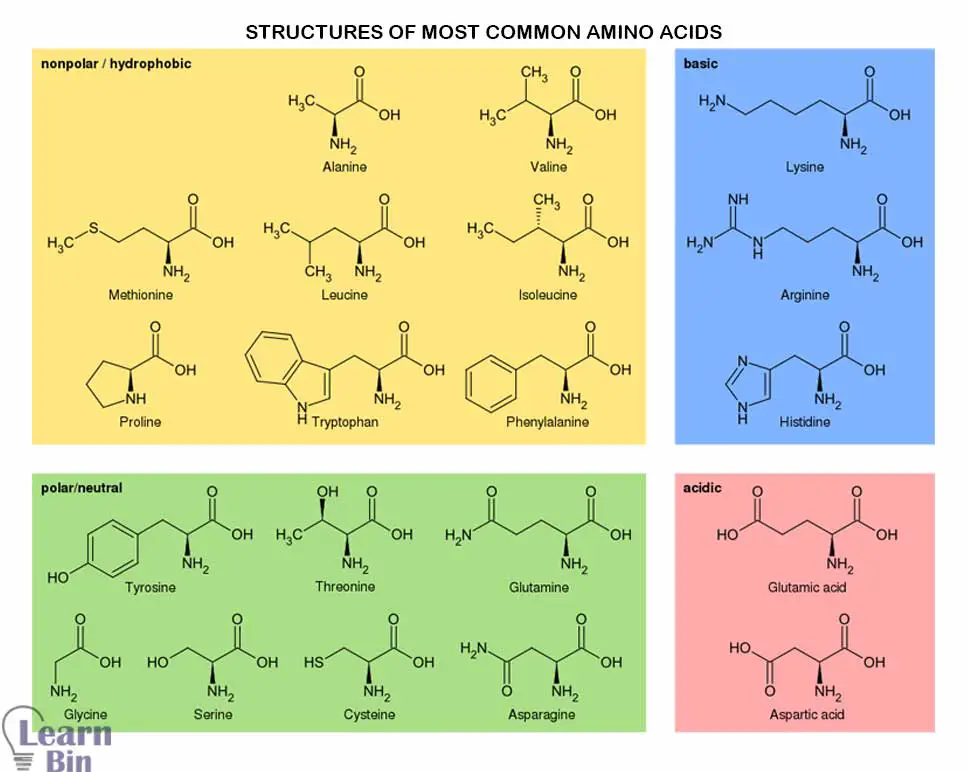
More than 500 amino acids have been found but only 20 amino acids appear in the genetic code. Of these 20 amino acids, 9 amino acids are essential amino acids. They cannot be synthesized in the body, so we need to get them from food.
| Amino acid | Abbreviation |
| Alanine | ala |
| Cysteine | cys |
| Aspartic acid | asp |
| Glutamic acid | glu |
| Phenylalanine | phe |
| Glycine | gly |
| Histidine | his |
| Isoleucine | ile |
| Lysine | lys |
| Leucine | leu |
| Methionine | met |
| Asparagine | asn |
| Proline | pro |
| Glutamine | gln |
| Arginine | arg |
| Serine | ser |
| Threonine | thr |
| Tryptophan | trp |
| Tyrosine | tyr |
| Valine | val |
Amino acids are bonded with each other amino acid molecules by peptide bonds. The carboxyl group (COOH) and amino group (NH2) of two different amino acids are reacted and release an H2O molecule. The hydroxyl group (OH) of the carboxyl group and a hydrogen atom of the amine group contributed to forming the H2O molecule.
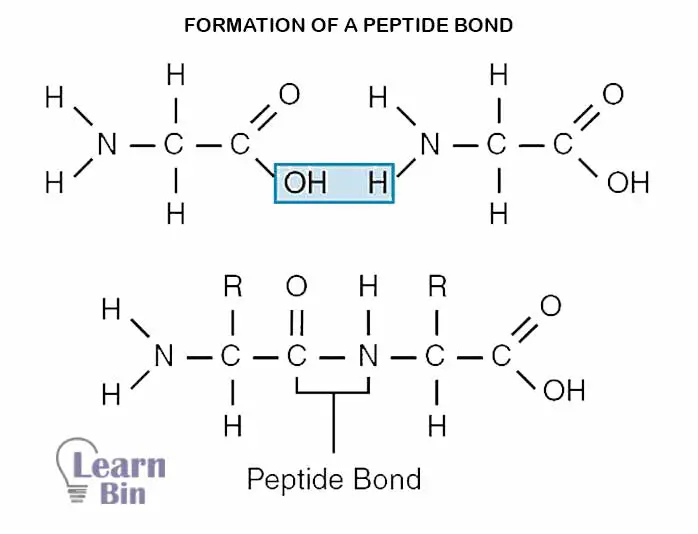
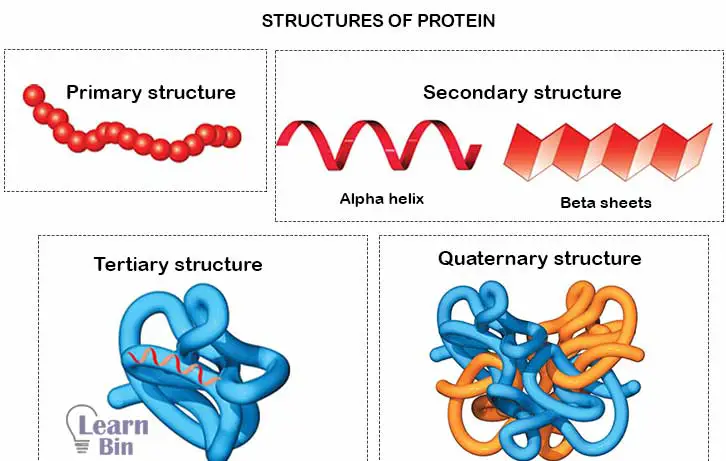
The sequence of amino acids in a linear polypeptide chain is called the primary structure. One end of the polypeptide chain has a Carboxyl group, and the other end has an Amine group. They are called carboxyl-terminal and amino-terminal respectively.
Examples:- insulin hormone
The secondary structure of a protein refers to the patterns of frequent bending of the polypeptide backbone. Hydrogen bonding between carboxyl groups and amino groups causes the polypeptide chain to bend. Alpha helix and Beta sheets are secondary structures of proteins.
Two polypeptide chains are bonded placed in an antiparallel direction and bonded by hydrogen bonds to form beta-sheets. In an alpha-helix structure, the polypeptide backbone is spiraling clockwise around an imaginary axis. A secondary structure can be known as an intermediate structure when forming the tertiary structure of the protein.
Examples:- keratin
Tertiary structure is the three-dimensional structure of proteins. The polypeptide chains are bending three-dimensionally and form 3D proteins. Hydrogen bonds, disulfide bonding, dipolar bonding, etc., contribute to forming the 3D structure of proteins. Enzymes and Antibodies are examples of the tertiary structure of proteins.
Examples:- enzymes ( amylase, maltase)
Several polypeptide chains are closely packed together and form the quaternary structure of proteins. Each polypeptide chains (subunit) in the quaternary structure have its own primary, secondary and tertiary structure.
Examples:- Hemoglobin
The breakdown of the 3D structure of the protein is called protein denaturation. Proteins lose their shape and properties when they denature. Protein denaturation is an irreversible process, and it is caused by the breakdown of hydrogen bonds between polypeptide chains.
Denaturation is usually caused by exposure to heat, extreme pH changes, saturated salts, exposure to radiation such as UV, γ - rays, X-ray, external stress, and heavy metals such as Ni, Cd, Cr, Pb, As, Ag, etc.
The relevant genetic instruction for protein synthesis appears in DNA. Synthesis of protein occurs on Ribosomes in the cytoplasm. The protein synthesis process happens in two steps.
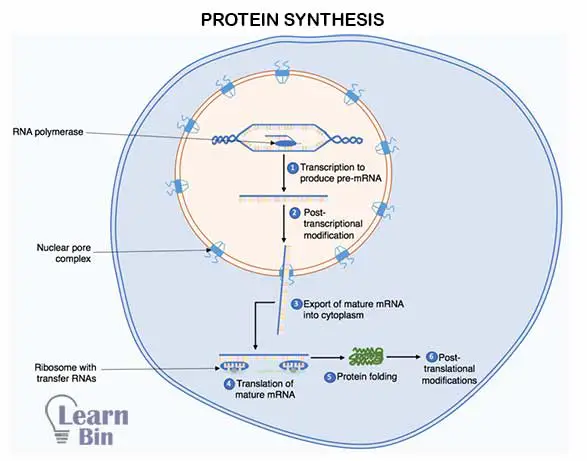
Here messenger RNA (mRNA) molecule is synthesized. First, a specific gene in a DNA molecule is unwinded. Then the hydrogen bonds between the two strands of DNA are broken down. Thus, the mRNA chain is synthesized as a transcript for one of the two separated strands of DNA.
There, an mRNA chain corresponding to the DNA chain is synthesized. This process is catalyzed by an RNA polymerase enzyme. The m-RNA chain leaves the DNA molecule and enters the cytoplasm through holes in the membrane of the cell nucleus. Then, the DNA molecule returns to its original state.
In the translation step, a polypeptide chain is synthesized based on the genetic information in the mRNA chain. The synthesized m-RNA chain binds to the ribosome in the cytoplasm. Ribosome identifies codons as base triads. When the ribosome reads the first codon in the mRNA chain, a tRNA molecule with the corresponding anticodon joins the corresponding amino acid molecule and binds to the ribosome.
Then the ribosome reads the second codon in the mRNA chain. The first amino acid is placed on the ribosome and the t-RNA that carries the first amino acid is removed. The second t-RNA molecule also comes with an amino acid molecule and binds to the ribosome. The first amino acid binds to the second amino acid in a peptide bond. Thus, the polypeptide chain is formed.
The codon is the three adjacent bases/ nucleotides in an mRNA chain or DNA chain that provides the code to determine a single amino acid in protein synthesis. The number of different codons that can occur from bases in DNA/RNA is 64. Three of these 64 codons are codons that stop the synthesis of protein. Those are known as stop codons.
3 stop codons:- UAG, UAA, and UGA
| Function | Example |
| Contributes to the formation of the structure of living organisms. | Keratin Collagen |
| Catalyzes biochemical reactions as enzymes | Maltase Lactase |
| Contributes to contraction and movements of the body | Actin Myosin |
| As animal hormones | Insulin |
| Provide immune protection as antibodies | |
| Contributes to the transferring of things through the body | Hemoglobin Fibrinogen |
| Important as storage of food | Caseinogen in mammalian milk Albumin |
| Act as a respiratory substrate | |
| As a toxin | Snake venom |
| Absorbs shocks | Albumin Intervertebral discs |

biology.arizona.edu - Large Molecules Problem Set
nature.com - Protein Structure
sciencedirect.com - Quaternary Structure
Cover Image was designed by Venita Oberholster from Pixabay
Figure 01: Techguy78, CC BY-SA 4.0, via Wikimedia Commons
Figure 02: original: Sponk, modified: isk, CC0, via Wikimedia Commons
Figure 03: OpenStax College, CC BY 3.0, via Wikimedia Commons
Figure 04: 4 organizations of proteins, CC BY 3.0, via Wikimedia Commons
Figure 05: Kep17, CC BY-SA 4.0, via Wikimedia Commons