More results...

Amino acids are the building blocks of proteins. So, the synthesis of amino acids happens naturally in our bodies. In the human body about half of the amino acids that are needed to make proteins, can be synthesized. Some amino acids are synthesized unnaturally/ artificially. These unnatural amino acids are more important in drug design for the pharmaceutical industry.
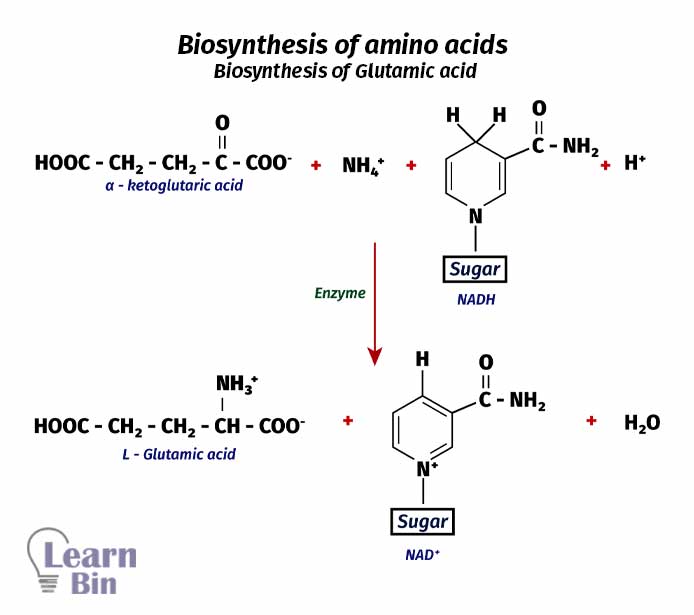
The biosynthesis of amino acids in our body is initiated with α-ketoglutaric acid. This reaction is a reductive amination reaction. First, NH4+ ions in our body are reacted with α-ketoglutaric acid in the presence of natural reducing agents like NADH. Here, the keto group in α-ketoglutaric acid will react with the NH4+ ion. This is an enzyme-catalyzed reaction. L-glutamic acid, NAD+, and water are the end products of this reaction.
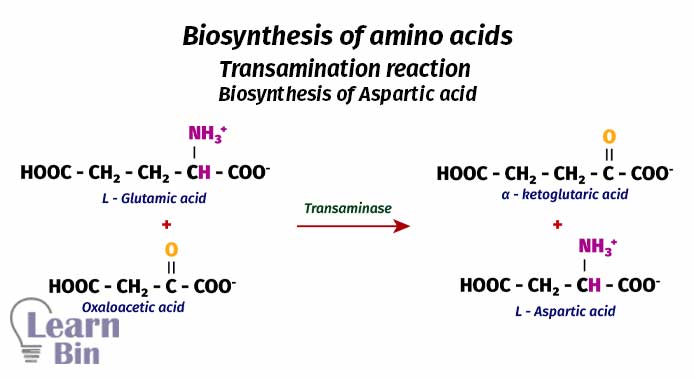
Using L-glutamic as the source, other amino acids are synthesized. To synthesize other amino acids, the amino group in the L-glutamic acid is transferred to another molecule. This type of reaction is called “Transamination”. This reaction is also catalyzed by enzymes. These enzymes are called “transaminases”.
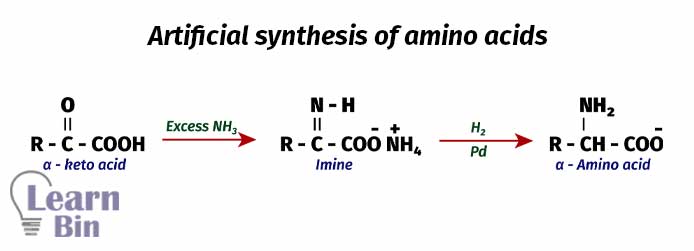
When the artificial or industrial synthesis of amino acid, first a keto acid is treated with excess ammonia. Here, the ketone group will react with ammonia and form an imine. Then the imine is reduced to an amine by Hydrogen and a Palladium catalyst. Under these conditions, the carboxylic group is not going to be reduced.
In natural systems, only L-type enantiomers are formed. Because a natural synthesis is initiated by enzymes. So, the natural synthesis of amino acids is a stereoselective reaction. But in artificial synthesis, both L and D enantiomers are formed equally. So, most of these reactions will produce Racemic mixtures.
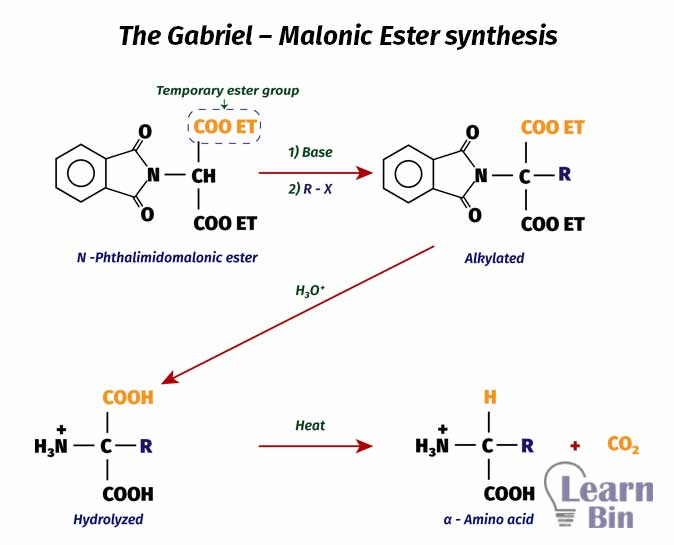
The Gabriel – Malonic Ester synthesis method is used to form amino acids that cannot be formed by direct amination of halo acids. This synthesis is started with an N-phthalimidomalonic ester. First, N-phthalimidomalonic ester is alkylated in a basic medium. After alkylation, acid hydrolysis is taken place to get an amine group. This reaction results in a dicarboxylic acid. By heating, one carboxylic group is removed as carbon dioxide. (Decarboxylation)
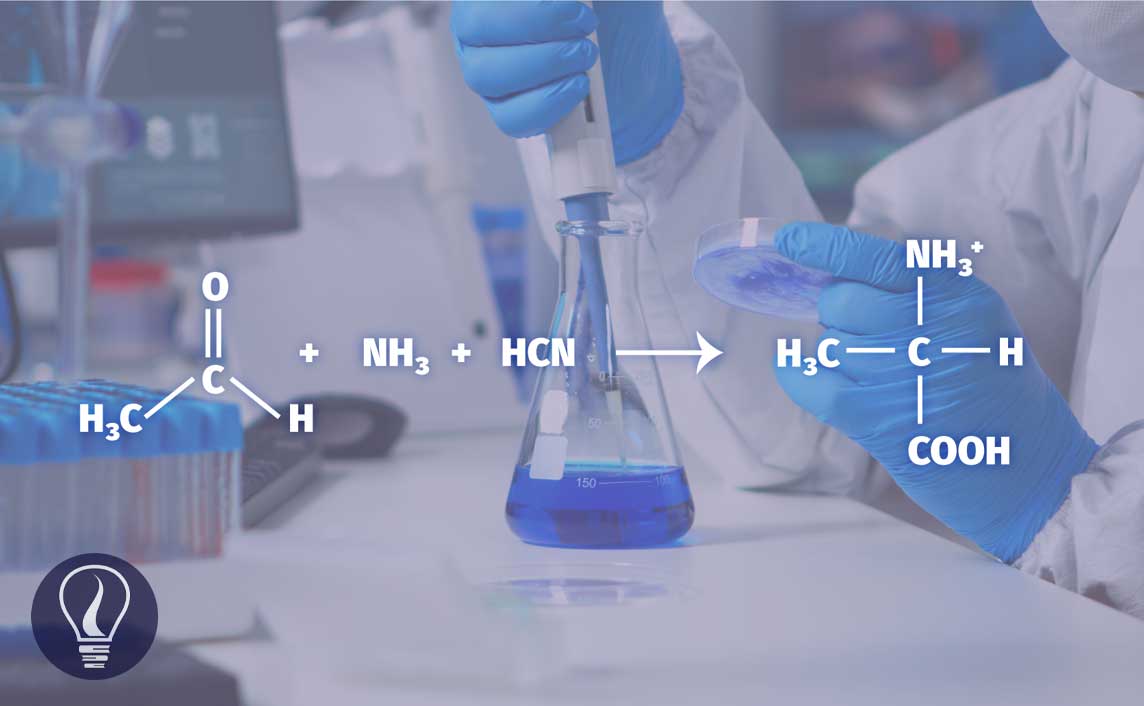
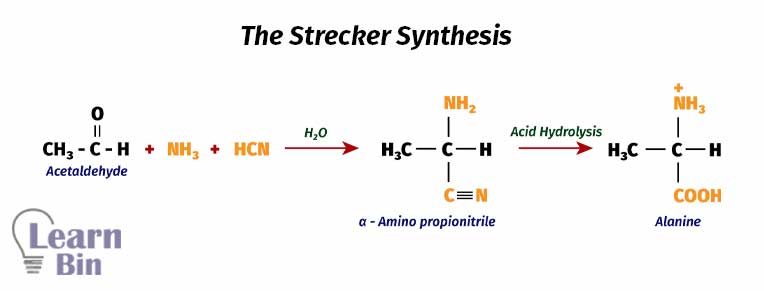
This is the most common method used in the industry. The Streaker method can be used to form a large number of amino acids from appropriate aldehydes. First, acetaldehyde is reacted with ammonia (NH3) and hydrogen cyanide (HCN) in a medium of water. This reaction results in α-amino propionitrile easily.
In this reaction, ammonia will react with the keto group and then the cyanide (-CN) group will attack it. Then the acid hydrolysis reaction takes place to convert the (-CN) group in α-amino propionitrile into a carboxyl group (-COOH). This reaction will result in a 60%/40% mixture of D-type and L-type alanine.

Amino acid biosynthesis - Deferrari, G., Mannucci, I. and Garibotto, G. (2010)
The cover image was created using an image by DCStudio on Freepik