More results...


The element nitrogen is a p-block element that belongs to the 15th group of the periodic table. The electron configuration of nitrogen can be written as N = 1s2 2s2 2p3. Nitrogen has a partially filled p orbital. It has 5 valance electrons. Either filling or removing all the electrons from the valance shell, nitrogen can show all the oxidation states from -3 to +5.
| The oxidation state of nitrogen | Compounds |
| -3 | NH3 - Ammonia NH4OH - Ammonium hydroxide NH4+ - Ammonium ion |
| -2 | N2H4 - Hydrazine |
| -1 | NH2OH - Hydroxyl ammine |
| 0 | N2 - Nitrogen |
| +1 | N2O - Nitrous oxide |
| +2 | NO - Nitric oxide |
| +3 | The oxidation state of nitrogen |
| +4 | NO2 - Nitrogen dioxide |
| +5 | N2O3 – Dinitrogen trioxide HNO2 - Nitrous acid NO2- - Nitrite ion |
The element, Nitrogen (N) exists as nitrogen gas (N2) in nature. Nitrogen gas is a colorless, odorless, neutral, and non-toxic gas. It partially dissolves in water.
The neutral behavior of the nitrogen gas is caused by its high bond energy. The bond energy of gaseous nitrogen is 945.33 kJ mol-1. This is because it has a triple bond between two atoms (two π and one σ bond). And gaseous nitrogen has the lowest bond length of 1.09 A°.
Ammonia is a colorless and toxic gas with a strong odor. In ammonia gas, there is a lone pair of electrons on the nitrogen atom. This lone pair is shared with Lewis acids and forms a dative bond. Therefore, ammonia possesses alkaline properties. It will easily dissolve in water and form the weak base of Ammonium hydroxide.
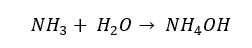
Ammonia has both reducing and oxidizing properties. Also, it has both acidic and alkaline properties. When ammonia reacts with elements that have low electronegativity (metals), ammonia acts as an oxidizing agent.
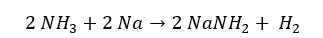
Here, hydrogen has reduced from a +1 oxidation state to a 0 oxidation state. And Sodium has oxidized into a +1 oxidation state. That means ammonia has acted as an oxidizing agent. Also, in the above reaction, Hydrogen is released.
If a compound reacts with a metal and releases hydrogen gas, the compound has acidic properties. The above example shows that ammonia gas shows acidic properties when reacting with low electronegative elements.
When ammonia reacts with the compounds with high electronegativity, it shows the reducing properties.
When ammonia is heated in the presence of oxygen gas, it will result in nitrogen gas and water vapor. Here, nitrogen has oxidized from -3 oxidation state to 0 oxidation state and oxygen is reduced to 0 oxidation state to -2 oxidation state. If the above reaction is done in the presence of Pt or Cu catalyst it will result in Nitrogen oxide instead of nitrogen gas. Here, nitrogen oxidized into a +2 oxidation state.
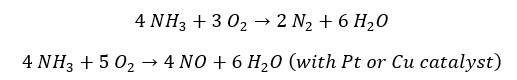
Excess of ammonia reacts with Cl2 as follows. By the reaction with Cl2, it forms HCl gas. Since there is excess ammonia it will further react with HCl and result in Ammonium chloride salt (NH4Cl)
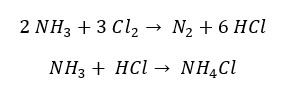
At the presence of excess Cl2, Br2 and I2, ammonia react as follows,
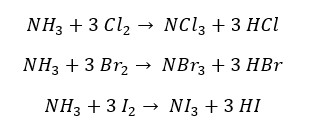
Ammonia reacts with acids and results in the respective salts. This explains the alkaline nature of ammonia.
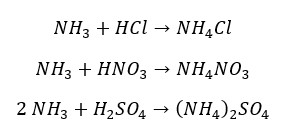
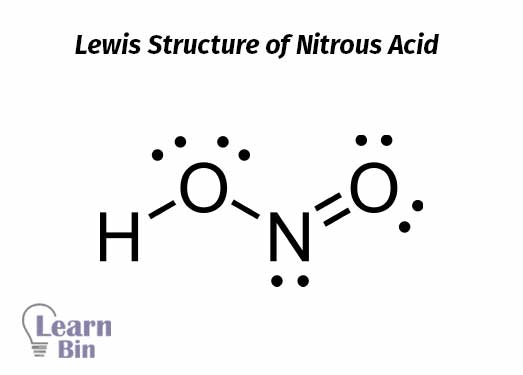
Nitrogen forms two oxo acids that are nitric(IV) and nitric(III) acids. Nitrous acid or nitric(III) acid is an unstable and weak acid. Nitrous acid can be obtained from the reaction between nitrites and dilute acids.
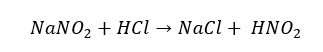
Nitric acid or nitric(IV) acid is a stable strong acid. Nitric acid is a strong oxidizing agent. Conc. nitric acid will degrade as follows and result in nitrogen dioxide, oxygen, and water.
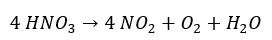
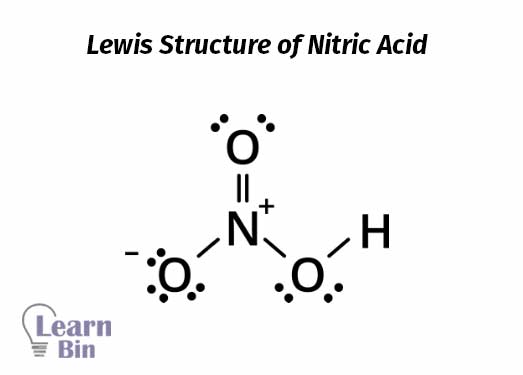

Figure 01: Contains an illustration by Calvero, licensed under Public domain, via Wikimedia Commons