More results...


Surface analytical techniques are specialized analytical techniques that use to gather information about the surface of a substance.
Most of those techniques involve bombarding the surface with a form of radiation such as electrons, photons, ions, or neutrons and then collecting the resulting emitted radiation (electrons, photons, ions, neutrons) using a detector.
The top layer of atoms (or molecules) of a substance that immediate interfere with other phases such as gas, liquid, or solid phases can be defined as the surface.
The structure and chemistry of that top layer of atoms (or molecules) are determined by the atoms or molecules immediately below that top layer. Therefore, the surface can be defined as the top 2 to 10 atomic or molecular layers of a substance. The thickness of the surface layer lies between 0.5 nm and 3nm.
In many applications, surface films are applied to materials to obtain different purposes such as,
Most of those surface films are in the range of 10 nm to 100 nm (could be much thicker in some cases). So, a depth of 100 nm from the top layer can be used to describe the surface because this area is responsible for determining the properties of the surface.
Beyond this 100 nm region, those layers are much more appropriate to describe the bulk solid state properties of the material.
Depending on this, the surface can be divided into 3 regions.
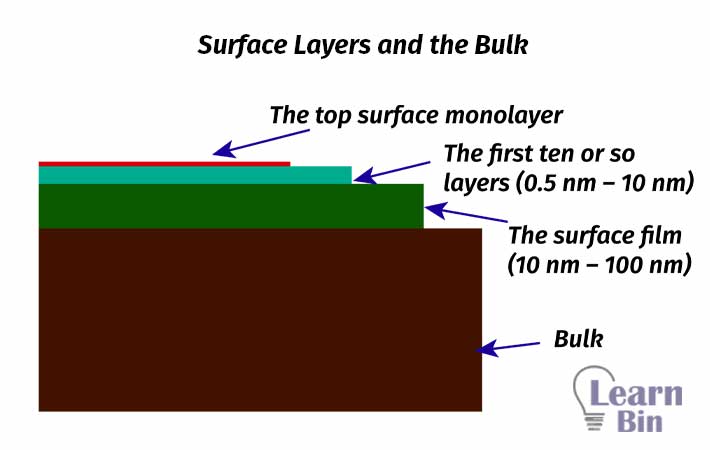
To study the surface of a solid material, surface analytical techniques should have the potential to distinguish the properties of all three layers of the surface as well as distinguish the surface from the bulk.
Let’s consider a metal cube of 1cm3 volume. It can be estimated that a surface that is 1cm2 contains 1015 atoms. So, within the 1cm3 volume of the cube, it can be estimated that there will be roughly 1023 amount of atoms.
When considering the surface-to-bulk ratio, can be calculated as follows.
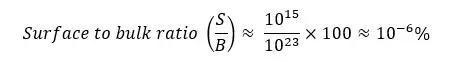
When looking at the above numbers it is obvious that the number of atoms (molecules) in the surface of the material is very small. So, examining the surface layer of atoms or molecules analytically and distinguishing the properties and the structure of the surface layer from the rest of the bulk material is not an easy process.
Most of the surface analytical techniques have the ability to analyze (probe) a surface area of 1mm2. Within this area, there are about 1013 amount of atoms. This can be considered a very low concentration when compares with other chemical analysis processes. To conduct that kind of analysis instruments and methods must be highly sensitive.
Things will get even more demanding and much higher sensitivity will be required when needed to analyze surface-influencing chemical species such as additives, and contaminants. Generally, those chemicals have much less concentration than surface atoms.
Let’s consider some other specific applications such as nanoparticle (nano surface) analysis, monitoring the distribution of catalyst across the surface of the support, monitoring homogeneity of a protective coating, monitoring optical property, and monitoring the effect of a drug on a cell. In most cases, those surfaces are in the micrometer range (some of them are much smaller than that). When that kind of analysis is required, that requires high spatial resolution.
So, conducting surface analysis demand higher surface resolution and sensitivity. To obtain that highly sensitive, specialized analytical instruments and equipment must be used.
The intention of surface analysis is to get an understanding of the properties, structure, composition, and reactivity of a surface. To obtain that, information about the surface can be collected using surface analytical techniques. These are the types of information obtained from the surface analysis.
There are many techniques to analyze surface as mentioned above. But all of the surface information cannot be obtained by one single method. To gather all the information about the surface, several different techniques must be used.
| Surface information | Surface analysis technique | Radiation in (provided) | Radiation detected |
| Physical topography | SEM | Electron | Electron |
| STM | Electron | Electron | |
| Chemical composition | XPS | Photon | Electron |
| AES | Electron | Electron | |
| SIMS | Ion | Ion | |
| Chemical structure | XPS | Photon | Electron |
| EELS | Electron | Electron | |
| Atomic structure | LEED | Electron | Electron |
| RHEED | Electron | Electron | |
| Adsorbate bonding | EELS | Electron | Electron |
| INS | Neutron | Neutron |
In most techniques, surface analysis processes must be conducted in a vacuum. The reason behind that is those techniques use energy sources (radiation) like electrons, ions, neutrons, and photons. If there are any molecules in the gas phase, they could interfere with the radiation (by reflecting, refracting, absorbing, or scattering radiation).
Also, to prevent surface contamination by the adsorption of particles in the gas phase, some instruments must be operated under ultra-high vacuum (<10−9 mmHg).
The surface analysis technique should have the ability to obtain information within the defined depth of the surface layer. A technique that has a good surface sensitivity should gather information from as close to the top surface monolayer. The surface sensitivity of an instrument is a measurement of the extent of the depth it collects the information.
In most methods, when the surface is bombarded with a source of radiation, the bombarded atoms or molecules will emit a form of radiation and detectors use this emitted radiation to gather information. When a surface is bombarded with radiation, that radiation has the ability to penetrate some layers of the surface. As a result, the detected radiation is generated in those deep layers. So when incident radiation penetrates the deeper layers, the information got from those methods has less surface specificity.
To obtain information about the surface, it is necessary to interfere with the surface somehow. As mentioned above, most techniques use bombarding the surface with a radiation source. That will affect the physical and chemical properties of the surface being analyzed. That will change the surface while the analysis happens. In most cases, the detected signal (radiation) could be interfered with by this surface damage.
It is important to understand this process because if not, the information generated from the surface could be a reflection of the surface damage (may not obtain the characteristic information of the surface).
Photon < Electron < Ion
| Radiation type (particle) | Penetration depth (Å) | Energy (eV) |
| Ion | 10 | 1000 |
| Electron | 20 | 1000 |
| Photon | 10 000 | 1000 |

Vickerman, J.C., and Gilmore, I.S. (2009) Surface analysis the principal techniques. ; John Wiley & Sons Ltd.
The cover image was designed using an image by SecretDisc, licensed under CC BY-SA 3.0, via Wikimedia Commons