More results...


Density is defined as the mass of a unit volume of any substance. If a substance has “m” mass and “V” volume, the density of that substance can be written as follows.
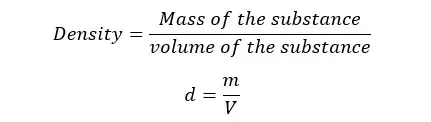
SI unit of density is kg m-3 (kilograms per cubic meter).
Density is constant for each substance. It only depends on the type of the substance. So density is an intensive property.
Examples of density test questions - Question 01
At 4℃, the density of pure water is 1000 kg m-3. Express the density of water in grams per cubic meter (g cm-3)
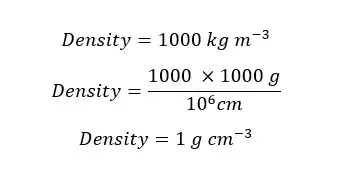
Examples of density test questions - Question 02
Find the mass of 500 cm3 of pure water at 4℃.
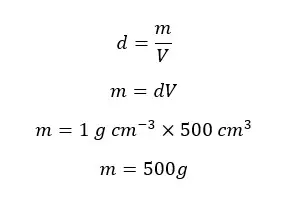
Examples of density test questions - Question 03
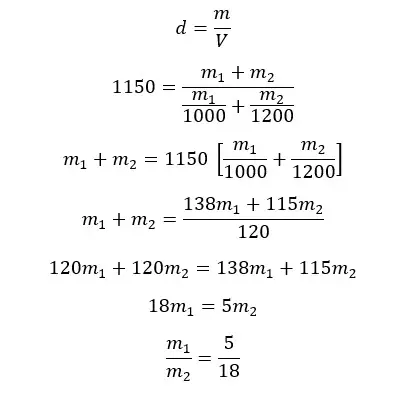
The densities of two liquids of A and B are 1000 kg m-3 and 1200 kg m-3 respectively. A homogeneous mixture is obtained by mixing A and B. The final mixture has a density of 1150 kg m-3. By considering there is no volume change when mixing, find the mass and volume ratios between A and B in the mixture.
Mass ratio
Let's take the masses of A and B are m1 and m2 respectively.
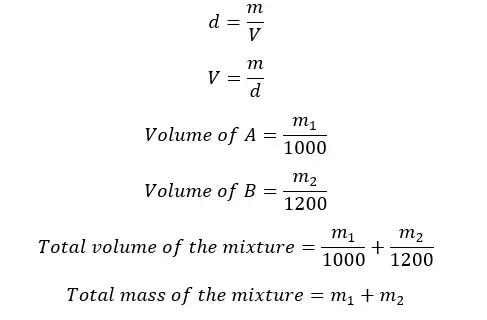
Let's apply the equation for density for the mixture.
Volume ratio
Let's take the volumes of A and B are V1 and V2 respectively.
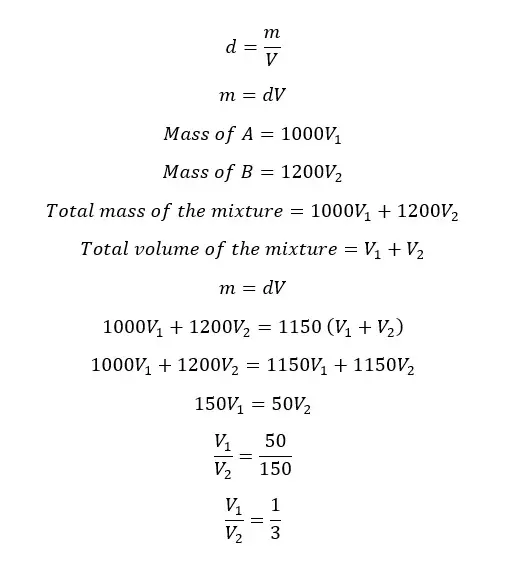
The ratio between the density of any substance, to the density of water is called the “Relative density”. Relative density is a unitless and dimensionless quantity.
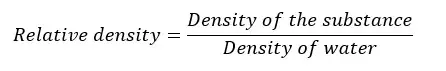
Examples of relative density test questions - Question 01
Find the relative density of coconut oil. (Density of coconut oil is 890 kg m-3 and the density of water is 1000 kg m-3)
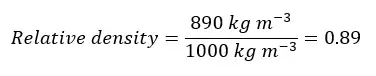
| Material | State of the material | Density (kg m-3) | Relative density (Also equal to density in g cm-3) |
| Water (at 40C) | Liquid | 1000 | 1.000 |
| Water (at 00C) | Liquid | 999.8 | 0.9998 |
| Water (at 200C) | Liquid | 998 | 0.998 |
| Hydrogen (at STP*) | Gas | 8.99 x 10-2 | 8.99 x 10-5 |
| Nitrogen (at STP) | Gas | 1.251 | 0.001251 (1.251 x 10-3) |
| Air (dry air at STP) | Gas | 1.275 | 0.001275 (1.275 x 10-3) |
| Ice (at 00C) | Solid | 917 | 0.917 |
| Sea water | Liquid | 1020 - 1035 | 1.02 - 1.035 |
| Blood (at 370C - average human body temperature) | Liquid | 1050.6 | 1.0506 |
| Diamond | Solid | 3515 | 3.515 |
| Magnesium | Solid (metal) | 1738 | 1.738 |
| Aluminum | Solid (metal) | 2700 | 2.700 |
| Iron | Solid (metal) | 7874 | 7.874 |
| Lead | Solid (metal) | 11340 | 11.34 |
| Mercury | Liquid (metal) | 13534 | 13.534 |
| Gold | Solid (metal) | 19300 | 19.3 |
| Platinum | Solid (metal) | 21450 | 21.45 |
| The core of the Earth | 9820 - 13300 | 9.82 - 13.3 | |
| Neutron star | Solid | 3.7 x 1017 - 5.9 x 1017 | 3.7 x 1014 - 5.9 x 1014 |
| The nucleus of an atom (average) | 2.3 x 1016 | 2.3 x 1013 | |
| Sun (average) | 1408 | 1.408 |
Also to calculate the density of a gas you can use 'The ideal gas equation'.

Where,

Table 01: Some data were referred from WolframAlpha.com
The cover image was created using an image by Zaccaria Boschetti from Pixabay