More results...


Temperature is one of the most important concepts in science. When describing characteristics of any system including the body of a living organism, mechanical system, chemical system, any substance, or planetary system, properties of that system are used. Among those properties, temperature plays an important role. The zeroth law of thermodynamics is responsible for the introduction of this important thermodynamic property, temperature.
But before the introduction of the zeroth law, people are familiar with the concept of temperature. Even thermometers were invented before the introduction of this law. That is why the zeroth law is known as the commonsense law. Even though people were familiar with this concept, scientists decided to introduce the concept as a law. When that happened, the first and second laws of thermodynamics were well established. But because the zeroth law is more fundamental than the other two, they assign it as the zeroth law.
If system A is in thermal equilibrium with system B and also system A is in thermal equilibrium with system C, then system B and C are in thermal equilibrium with each other.
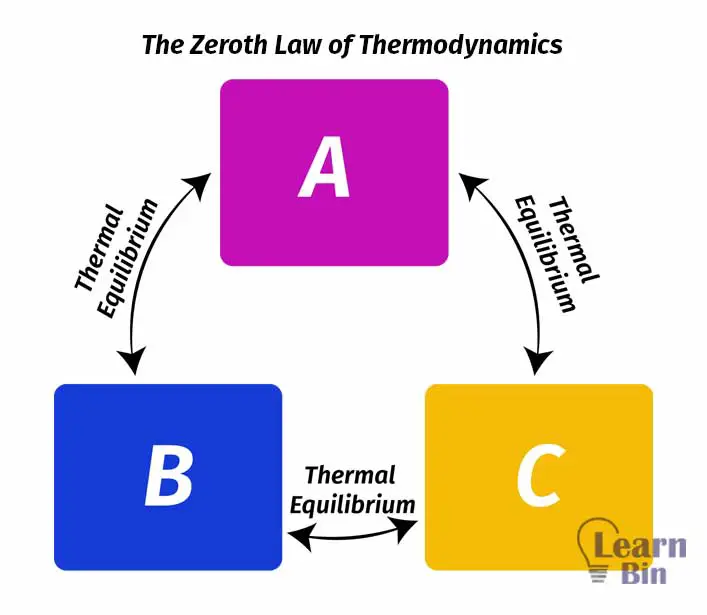
The above concept is known as the zeroth law of thermodynamics.
Simply equilibrium means, the state of balance between two or more systems. That means those systems will be in a stable and balanced state.
If no change in any macroscopic properties is observed in a system that is isolated from its surrounding, that system is in a state of thermodynamic equilibrium. An isolated system always reaches the state of thermodynamic equilibrium over time and that system never leaves that state of thermodynamic equilibrium spontaneously.
For a system to be in the state of thermodynamic equilibrium, the following conditions should be satisfied.
For a system to be in mechanical equilibrium, all the forces within the system and between the system and its surrounding are balanced. If there are any unbalanced forces present, either the system alone or both the system and its surrounding undergo change until the system reaches the mechanical equilibrium.
For a system to be in chemical equilibrium, there shouldn’t be any chemical reaction (the net amount of reactants and products should be constant) or shouldn’t be any matter transfer (ex: diffusion) within part of the system.
A system that is surrounded by a diathermic wall, exists in states of both mechanical and chemical equilibriums and if that system shows no spontaneous change in any property of the system, that system has attained the state of thermal equilibrium.
Any system that failed to satisfy any of the above three conditions, that system is in a non-equilibrium state.
The zeroth law is the basic concept behind the measurement of temperature. Temperature is the thermodynamic property of a system that helps to determine that system is in thermal equilibrium with other systems. A reference body is used to get a quantitative measurement of temperature. This reference body should have distinguishable physical characteristics/properties that change with the temperature.
This reference body used to measure temperature quantitatively is called a ‘thermometer’. The property that a thermometer has those changes with temperature is known as the ‘thermometric property.’
The thermometer is important to measure the temperature of a system. When a thermometer is placed contacting a system that is needed to measure temperature, the thermometer, and the system will be reached thermal equilibrium. So, the temperatures of both systems will be equal. Using the temperature scale in the thermometer, temperature readings can be observed.
So, by observing the change of this thermometric property to changing temperature, the temperature can be measured quantitatively. When selecting a good thermometric property for observing temperature reading accurately, it should have some important qualities.
Using this concept, there are many types of thermometers are available.
| Thermometer | Thermometric property |
| Thermocouple | Electromotive force |
| Electrical resistance thermometer | Resistance |
| liquid-in-glass thermometer | Expansion of liquid (change in length) |
| Constant pressure gas thermometer | Volume |
| Constant volume gas thermometer | Pressure |
To assign a temperature value for a body/ system, a scale for the temperature is required.
Let’s find out how to measure temperature using changes in thermometric properties in thermometers.
The temperature of the system, θ is a function of thermometric property, X. So, let’s indicate this function as θ(X), and also let’s assume that the function is linear.
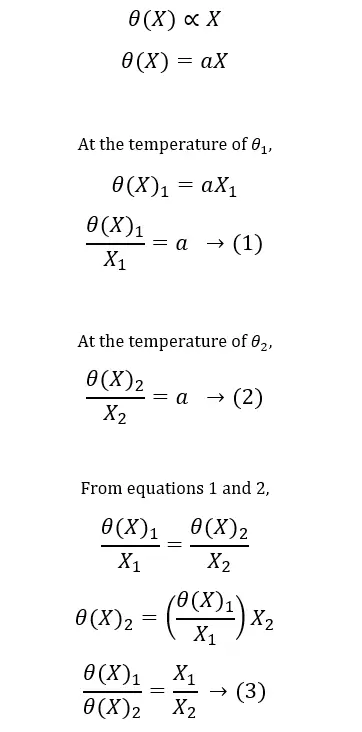
Before 1954, two fixed points were used to calibrate a thermometer. Those points were the ice point of water and the steam point of water.
A fixed point in a temperature scale means, ‘the value of temperature that is assigned to an easily reproducible state of a system’.

But after 1954, only one fixed point is used because the two fixed points that were used before are not reliable.
As a solution for this, the triple point of water has been introduced as a fixed point. The triple point of water is the state where ice, liquid water, and water vapor coexist in equilibrium. So, since 1954, only one fixed point is used to calibrate thermometers. The value of this point is assigned as 273.16 Kelvin (273.16K) (relative to the absolute zero – 0K). Because this fixed point is easily reproducible, the triple point (tp) of water is the standard fixed point of thermometry.
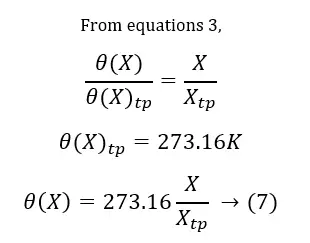
The above principle can be applied to measure temperatures using different thermometers. (Table 02)
Using the below equation temperatures can be converted between these two scales.
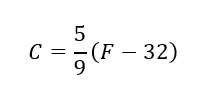
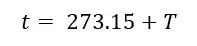
If a temperature of a given system was measured by using a few different types of thermometers at the same time, readings obtained from them will be different by a considerable amount. The smallest variation between readings will be observed from gas thermometers. So that is the reason behind choosing gas as the standard thermometric substance. So, because of its highly accurate measurements, the Constant Pressure Gas Thermometer is used to calibrate other thermometers.
| Thermometer | Symbol of the Thermometric property used | Temperature measurement (θ) according to equation 7 θ = |
| Thermocouple | ε (Electromotive force) | 273.16(ε/ εtp) |
| Electrical resistance thermometer | R (Resistance) | 273.16(R/ Rtp) |
| liquid-in-glass thermometer | L (change in length) | 273.16(L/ Ltp) |
| Constant pressure gas thermometer | V (Volume) | 273.16(V/ Vtp) |
| Constant volume gas thermometer | P (Pressure) | 273.16(P/ Ptp) |
When making a good thermometer there are many important concepts involved such as the thermal expansion of solids, liquids, and gases, the concept of ideal gases, the effect of the temperature on resistance, and electromotive force. They will be discussed in different articles. Also, the concepts behind different thermometers will be discussed differently.

Atkins, P. Four laws that drive the universe; Oxford University Press: Oxford, 2007.
Nag, P. Basic and applied thermodynamics; Tata McGraw-Hill: New Delhi, 2010.
The cover image was created using an image by Mudassar Iqbal from Pixabay