More results...


Latex dipping is a technique used in industries based on the manufacturing of thin walls or thin-film latex products. Mainly gloves, balloons, condoms, masks are manufactured by the latex dipping process. Simply molds or formers are dipped in the compounded latex and form a thin layer of the latex. The thickness of the deposit produced can be altered by changing various parameters such as viscosity of the compound, dwell time, number of dipping, and use of a coacervate, etc.
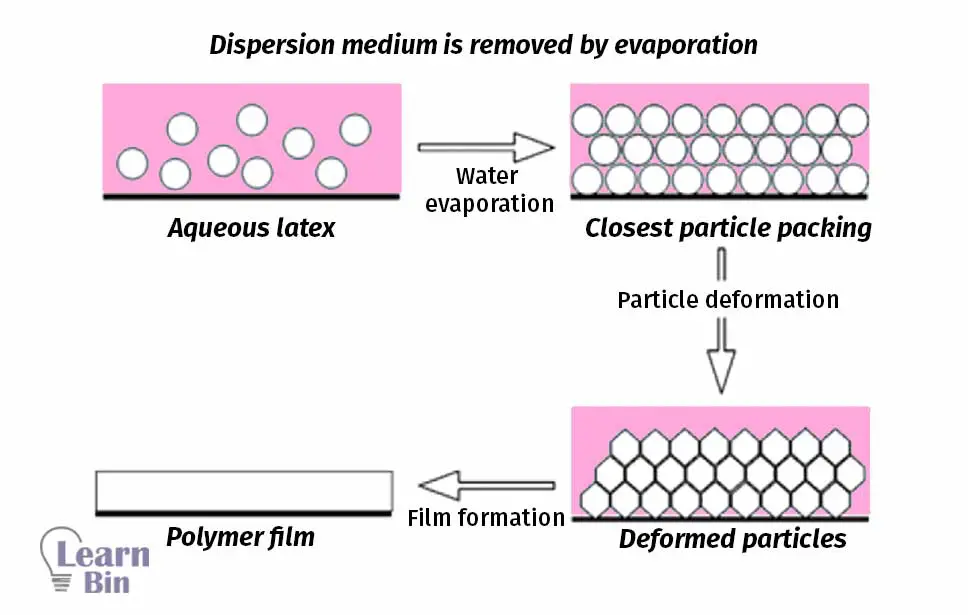
The compounded latex is still in wet form, and it is stabilized in its dispersion medium (water). When making products, the dispersion medium should be removed. In other words, the electrostatic stability of the latex is collapsed, and coagulation occurs. (Latex destabilization). The latex destabilization can be carried out by either physical or chemical methods.
In physical destabilization methods, the dispersion medium is removed by heating, freezing, evaporation, or mechanical agitation.
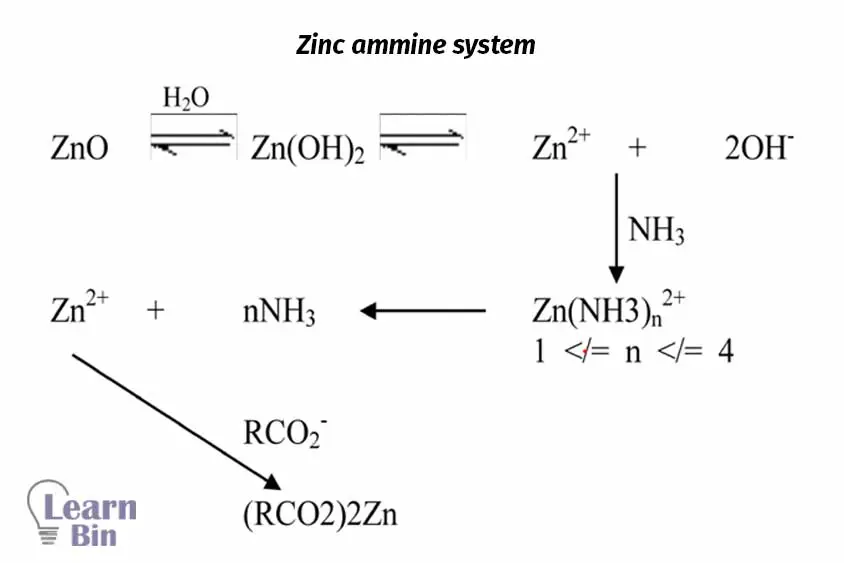
In the chemical method, specific coacervates are used. this can be described under three groups.
Materials that would bring about immediate destabilization as soon as they are added to NR latex. Therefore, the direct coacervates do not add to the latex compound. The mold or former is first dipped in direct coacervates and then dipped in compounded latex.
As the instant destabilization of latex, a thin layer of latex is obtained. Acidic substances, metallic ions, cationic surfactants, water-soluble organic matrix, and rubber soluble organic matrix are used as direct coacervates.
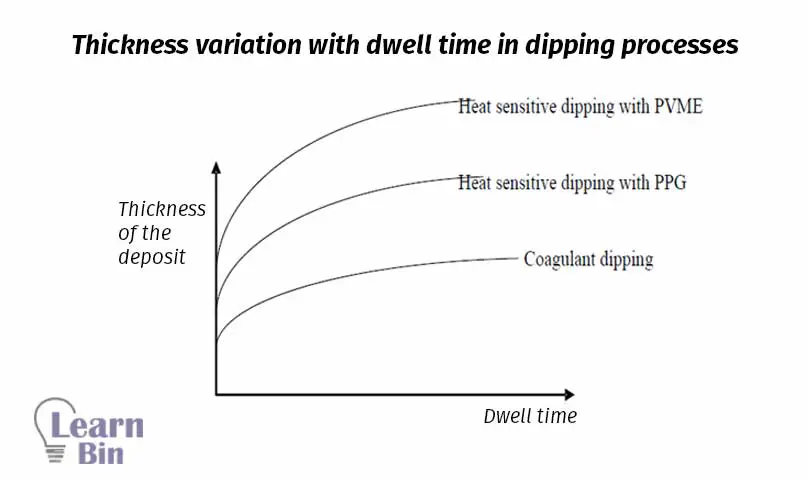
Heat sensitizing coacervates destabilize the latex when they are applied heat. These coacervates are added to the cold latex compound. The mold or former is first heated and then dipped in the latex compound. Therefore, on the mold, a thin layer of latex film is formed. Three principal systems can be used for heat sensitization of NRL.
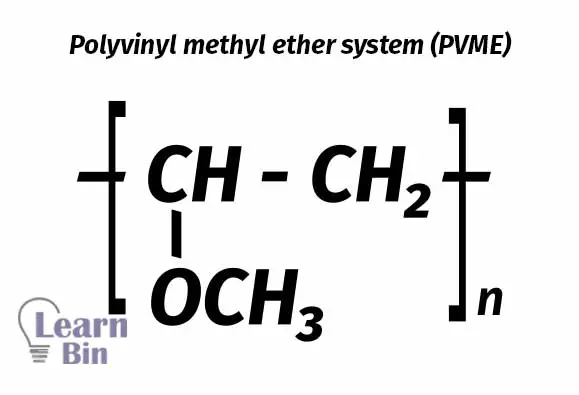
PVME is soluble in cold water but insoluble in hot water. The ability of PVME to heat sensitize the NR latex is a result of this behavior.
PPG is also a non-ionic hydrophilic polymer soluble in cold water, but insoluble in hot water as PVME. The mechanism of heat sensitization given by PPG is related to this solubility behavior.
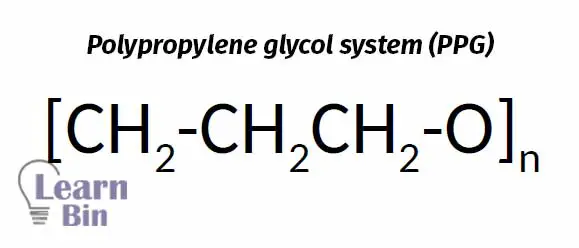
Delayed action coacervates take some time to take place in the destabilization process. In the foam manufacturing industry, delayed action coacervates are widely used.
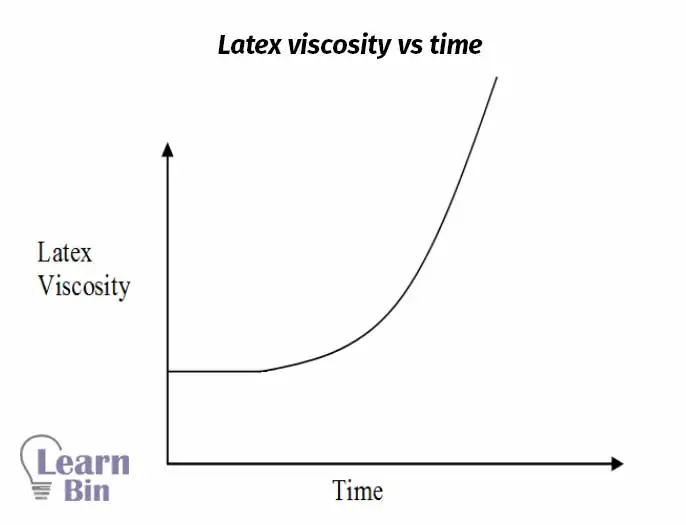
Straight dipping also known as simple dipping is carried out without the assistance of a coagulant (coacervates). The formation of the deposit depends on two factors which are uniform wetting of the former by the latex and the viscosity of the latex. The thickness of the rubber deposit obtained in one straight dip is usually around 0.05 mm.
Widely used technique in the dipping industry. The principal advantage of this process is, it produces considerably thicker deposits than the simple dipping process. The thickness of the deposit obtained in one coagulant dipping is around 0.2 – 0.8mm. The process can be divided into two methods.
Wet coacervates such as acetic acid are used. This can be done in two ways.
The former is first dipped in the coacervate
This type of wet coagulant dipping is not widely used. The disadvantages of this process are the non-uniformity of the deposit and the tendency of the deposit to slip off the former during the withdrawn.
This slippage is due to the liquid layer in between the deposit and the former. The liquid layer formed partly from the wet coagulant and partly from serum.
The former is first dipped in the latex compound
The mold is first dipped in the latex compound and then withdrawn. Next, it is dipped in the coagulant and withdrawn. Again, the mold is dipped in the latex. The former is allowed to dwell in the latex for a predetermined time to get the required thickness of the deposit. One disadvantage of this second method is that it involves two latex dipping.
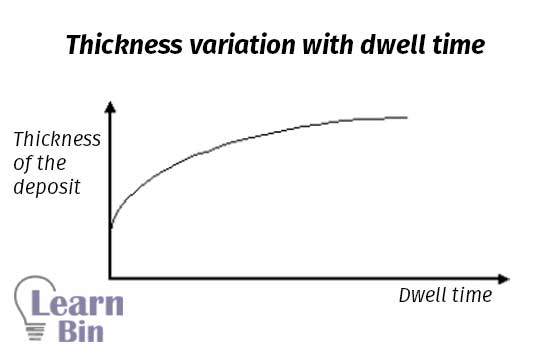
The most widely used method in coagulant dipping is the dry coagulant dipping method. In the latex industry, CaCl2, Ca(NO3)2 are used as dry coagulants.
It is possible to further layers of rubber formed by repeated coagulation and latex dipping. This multiple dipping method produces thicker films. Also, it is possible to make films containing more than one type of rubber. Sometimes, the inner layer may be produced from NR to take advantage of elasticity and strength whereas the outer layer may be produced from a synthetic rubber that has higher oil and chemical resistance.
| Ingredients | Parts by weight |
| CaCl2 | 15 |
| Ca(NO3)2 | 15 |
| (IMS) industrial methylated spirit | 50 |
| Water | 20 |
| Wetting agent (nonionic surfactants) | 0.001 (additional) |
In addition to the above salts parting agents such as CaCO3, talc at the level of 1.5% by weight of the level of coacervates can be used to facilitate subsequent dry deposit on the former.
A heated former and a heat-sensitive compound is used. The thickness of the deposit depends on the degree of heat sensitivity of the latex compound, the temperature of the former, and the heat capacity of the former. The most widely used heat sensitivity agent is PVME. A single heat-sensitive dip can produce a latex thickness of up to 4mm. This process is used in the production of thick-walled articles such as baby teats and electrician gloves.
| Compounding ingredient | Parts by weight |
| Latex 60% | 100 |
| KOH 205 | 0.2 |
| Potassium laurate 20% | 0.4 |
| Sulfur 35% | 1.2 |
| ZnO 50% | 1 |
| Accelerator 50% | 1 |
| ZMBT 50% | 0.2 |
| Antioxidant 50% | 1 |



The cover image was designed by using an image by Marketingdepascale, licensed under CC BY-SA 4.0, via Wikimedia Commons
Figure 10: Image from balloonhq.com - How Balloons are Made
Hello Mam, We are manufacturers of Industrial and household rubber gloves currently, and looking to manufacture Electrical Insulating Gloves, we required compounding Ingredients formulation, list of insulation fillers and solutions and Process of Manufacturing step by step on Our Batch type Dipping systems.
Hello, actually i am working in a latex glove factory as a R&D engineer, i would like to get in contact with you to know if you can help me. I will send you an e mail to [email protected] expleaning my questions. Thanks Julio
Can I have a contact method of saumya madhubashini?
We cannot share our personal contact information. But please send an e-mail to [email protected] by mentioning your concern / your problem.
Can I have saumya madhubashini email.
Iam into latex extrusion
We cannot share our personal contact information. But please send an e-mail to [email protected] by mentioning your concern / your problem.
I would like to set up a small Latex Dipping Center in Singapore for Heat Sensitive for Dipping Latex for engineering parts. I would like to know if you have any more details on this. The article above has given me some insight but would like to get more details on it. The thickness I am looking is a 1.2 to 1.5 mm
Dear madam,
I am working in the Quality Assurance section of a latex glove unit having previous experience in latex centrifuging.Today I happened to read your articles related to rubber latex technology.I am very much interested about this topic and learned more about latex technology.In the dipping process you have peripherally mentioned common defects formed during dipping process.It would be helpful for us to get a detailed articles related to common defects during dipping process that means the rootcause for the defects and remedies to overcome the defects so as to get good quality gloves . Moreover it will be useful for us if we get more informations,like how the latex quality influences the product quality from you. Thank you very much.
Thank you very much for your honest feedback.