More results...


Polymers are mixed together and processed in order to form polymer blends. Most of the polymer blends show phase separation after mixing. These types of blends are called immiscible polymer blends. The polymer blend can be miscible, immiscible, or partially miscible depending on the free energy.
ΔGmix = ΔHmix – TΔSmix
Mixtures are systems that contain two or more different chemical species. Binary mixtures are consisting of two different species (PS and Polybutadiene). And ternary mixtures are consisting of three different chemical species (PS and Polybutadiene in toluene)
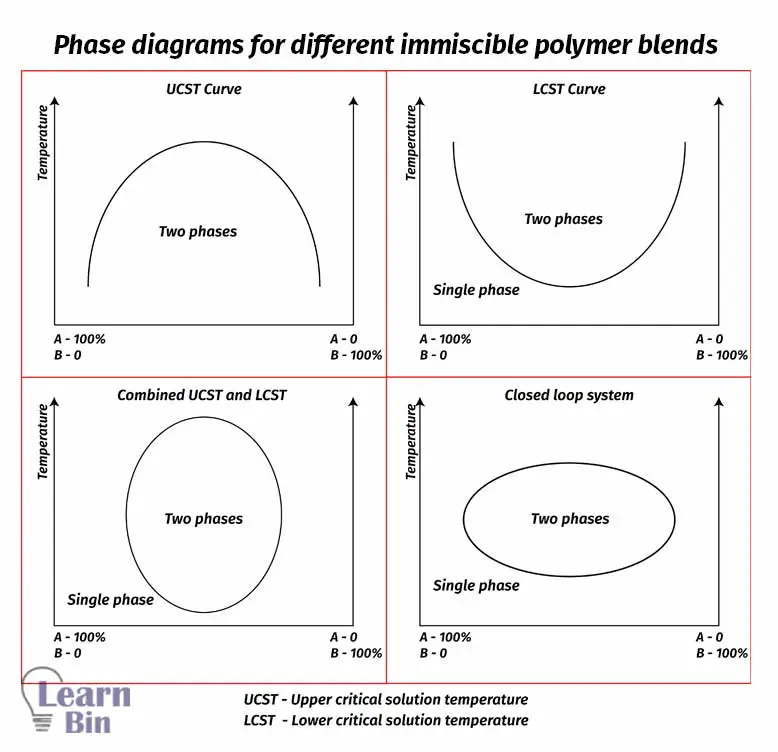
If the mixture is uniform and all components of the mixture are intermixed on a molecular scale, the mixture is called homogeneous. If the mixture consists of several different phases (regions with different compositions) it is called heterogeneous mixtures. For the miscibility of a binary blend, the following two conditions must be satisfied.
ΔG < 0
and
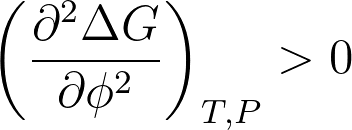
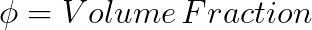
If the mixing is exothermic then the system is driven toward miscibility. The mixing is exothermic only when strong specific interactions occur between the blend components. The most common specific interactions found in polymer blends are hydrogen bonding, dipole-dipole, and ionic interaction.
To occur a phase separation ΔGmix should be greater than zero. Enthalpy of mixing (ΔHmix) plays an important role in phase separation. If the ΔHmix is less than zero, the mixture is stable and no phase separation occurred. When ΔHmix = zero, the system is in a stable state due to favorable entropy. This is the ideal state. In an ideal system ΔGmix = – TΔSmix.
When ΔHmix is slightly greater than zero, the system is still stable state and has no phase separation. Because entropy dominates. If the ΔHmix >> 0, enthalpy dominates. In such systems, phase separation occurs. In all cases as temperature (T) increases, entropy contribution becomes more important, so at very high temperatures mixing is favorable. But in polymer blends, increasing the temperature is not suitable to obtain immiscible blends. Because at high temperatures (2000K – 3000K) polymers will degrade.
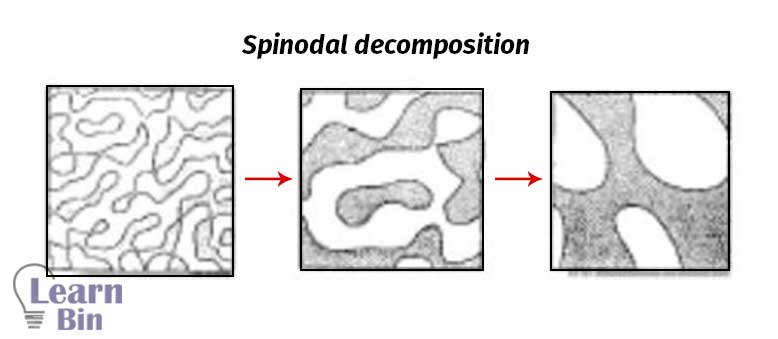
Spinodal decomposition is a spontaneous process. Therefore, there is no thermodynamic barrier to this process. The system is miscible at an early stage and due to the fluctuation of free energy, phase separation immediately occurs.
In some blends, small polymer droplets which are called nuclei are formed. Then this nucleus grows as a phase-separated domain.
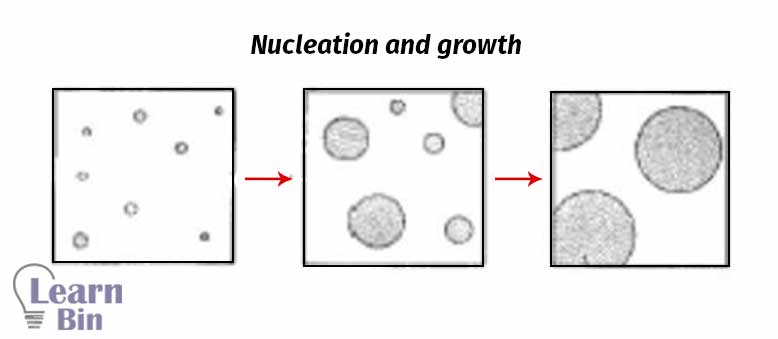
A binary system of polybutadiene and polystyrene is an immiscible blend. Two polymers are separated from each other and polybutadiene is arranged into little spherical shape blobs. These polybutadiene spheres affect the properties of the blend. PS is a rather brittle material. It is stiff but brittle.
But those little polybutadiene spheres are rubbery and can absorb energy under stress. Therefore. PS won’t break. This immiscible blend has more ability to bend instead of break than regular PS. Now the blend is tougher and more ductile. This PS and Polybutadiene system is commercially available as HIPS.
Another example of immiscible blends is polyethylene terephthalate (PET) and polyvinyl alcohol system (PVA). PET and PVA are immiscible and separate into sheet-like layers called lamellae. This blend is good to make plastic bottles for carbonated beverages. The PET makes the bottle strong, while the layers of PVA block the diffusion of CO2.
The blending of polymers results in materials that have some of the properties of one polymer, and some of the properties of another. Mixing two or more polymers together to form a blend that will hopefully have some properties of both in the right combination.
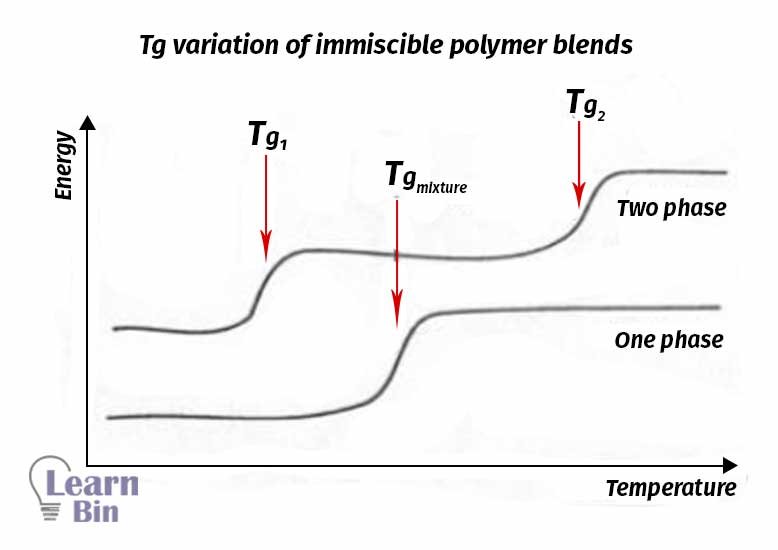
Adding incompatible components enhances the free volume of the system and hence lowers the Tg. The behavior is explained by Fox Equation. The blend has Tg in between the Tg values of both polymers.
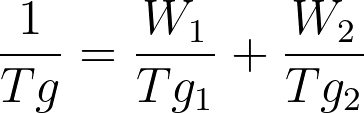
W1 & W2 = Weight fractions of two components
Tg1 & Tg2 = Glass transition temperatures of two components
Tg = Glass transition temperature of the polymer blend
In some immiscible blends, minor components give reinforcement to the material. Minor components which form spherical shape blobs in the matrix, form rod-like structures when processed underflow. These rod-like structures act like fibers and they give the material extra reinforcement.
Another way to make a strong immiscible blend is to use more equal amounts of the two polymers. When the relative amounts of the two polymers are equal, we get a different morphology (co-continuous phase) than when one is in large excess. This means both phases will be bearing a load of any stress on the material, so it will be stronger.

The cover image was created using Image by Carol L Simmons from Flickr