More results...


Synthetic rubbers are man-made lattices. Unlike natural rubber, we have no raw materials to make synthetic rubber. Synthetic rubbers are made by polymerizing particular monomers. Synthetic lattice productions have high demand because they do not have any protein allergy issues.
However synthetic rubbers cannot give as much elasticity as natural rubber. Chemical resistance properties are also given highly by synthetic rubbers. Special grades like Nitrile rubber and butyl rubber are from synthetic rubbers. Synthetic rubbers are mainly co-polymers which have two types of monomers. These two monomers are able to make co-polymers as well as homopolymers.
The majority of synthetic lattices of industrial importance are co-polymers. This is because we can have intermediate properties by mixing homopolymers and manufacturing co-polymers.
Styrene-butadiene copolymer lattices form a large family of general-purpose synthetic rubbers which are industrially very important. There are several grades of SBR due to different manufacturers and different uses. According to the production process, different grades of SBR can be produced with different ratios of styrene and butadiene. The styrene: butadiene ratio of the dispersed polymer has at least five important effects on SBR copolymer latex and the polymer itself. Which are,
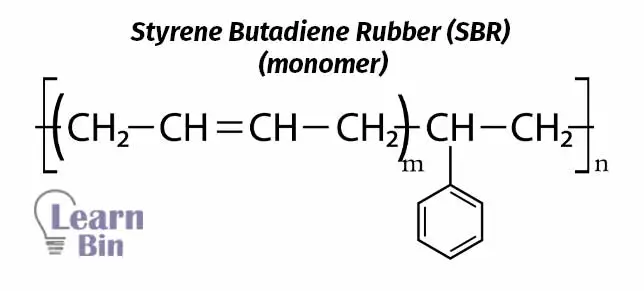
With the increase of the ratio of butadiene, the flexibility of the copolymer is increased by increasing the crosslinks. So, to increase the rubbery properties, it is essential to increase butadiene content. Increasing the styrene content increases the resistance of the copolymer to degradation. This is because styrene has unsaturated carbon rings which have high stability. The resistance is decreased when increasing the unsaturation by increasing the butadiene percentage.
When styrene is increased by more than 70% it will deposit as a powder. So, it is hard to get a uniform coherent. Therefore 70:30 styrene: butadiene ratio is the most suitable grade. If the styrene: butadiene ratio is less than 70:30 the latex readily foams a coherent film when drying at normal temperatures. The tensile strength and elongation at the break of un-vulcanized latex films also vary with the styrene content.
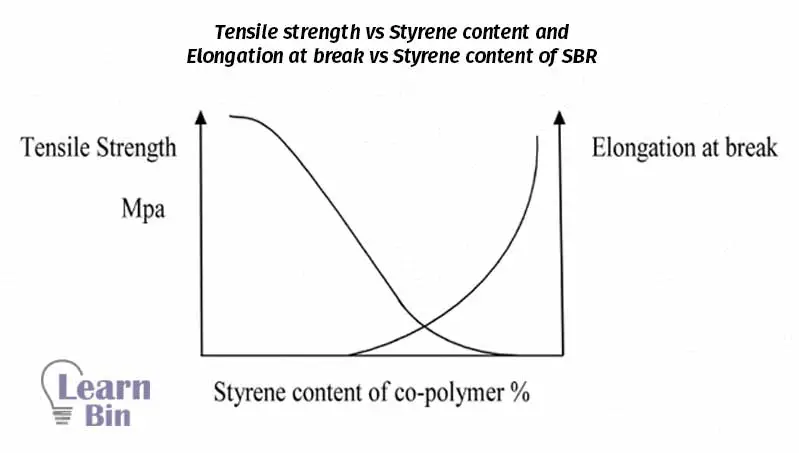
The styrene-butadiene ratio also affects the chemical properties of SBR co-polymers. As the styrene percentage is increased the concentration of un-saturation in the copolymer decreases. Therefore, the reaction; is Sulphur vulcanization is retarded. At the same time, the resistance of copolymer to deterioration by ozone and oxygen is improved as styrene content is increased.
The extent of branching and cross-linking of the polymer during the synthesis is also affected as this reaction depends on the remaining un-saturation of polymerized butadiene units. The lower the styrene percentage is the greater the tendency for branching and cross-linking.
The colloidal stability of SBR copolymer rubbers also depends on the butadiene monomer ratio. The MST of latex decreases with decreasing S: B ratio from 100/0 -to 50/50. Styrene particles are hard glassy particles. They are crystal-like materials. Such particles are less likely to coalesce when they collide because of mechanical agitation.
Some of the important applications for SBR lattices are,
They can also be used as stiffening and reinforcing agents for films of rubbery lattices. The physical character of the film derived from SBR lattices changes progressively as the styrene content is increased. Lattices of copolymers of relatively low styrene content are used as an alternative to NR latex. SBR copolymer lattices used as binders in latex-based paints have rather higher styrene content.
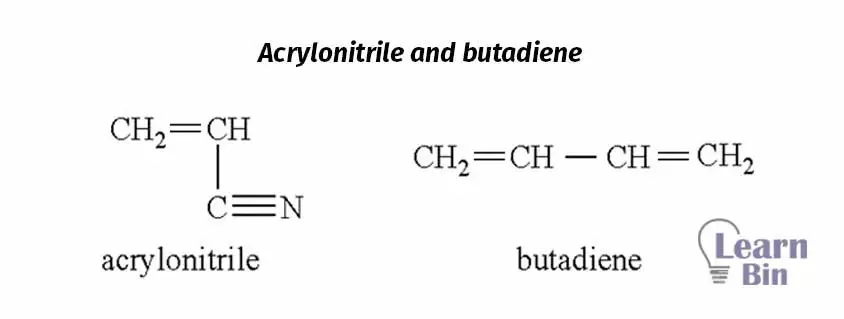
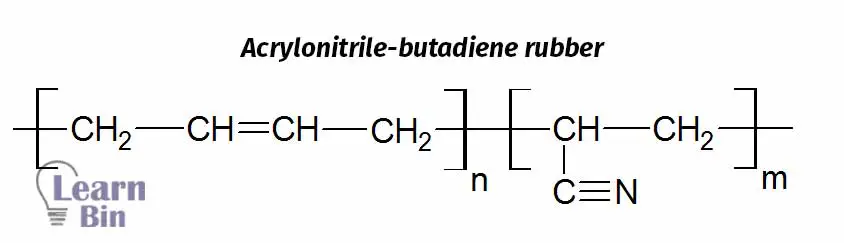
Acrylonitrile butadiene rubber is produced by emulsion copolymerization of Acrylonitrile (ACN) and butadiene. The Glass transition temperature (Tg) of polyacrylonitrile is about 90 ℃ and the Tg of polybutadiene is about -90℃. As the ACN/butadiene ratio increases the polymer changes from a rubbery material to a glassy material. So, rubbery films can be obtained by maintaining the ACN content is less than 50% of the copolymer lattices.
The repeating units of the acrylonitrile are polar due to the CN group. The increase in Tg with increasing ACN is due to the polarity of these units. Apart from increasing Tg it also increases the adhesion to polar substances. Because of the polar groups, Acrylonitrile gives the co-polymer its oil-resistant properties. Also, it gives chemical resistance to the polymer because most of the chemicals are non-polar.
Commercially three types of Nitrile Butadiene Rubber are available. They are classified as low, medium, and high nitrile latex.
| Rubber grade | Nitrile content |
| Low | 25% |
| Medium | 33% |
| High | 45% |
Polychloroprene rubber can be obtained by emulsion polymerization of chloroprene. Polychloroprene is a polar rubber. It is more polar than polybutadiene but less polar than poly-acrylonitrile butadiene copolymer lattices. Tg is around -45℃. The only difference between polychloroprene and natural rubber is, that polychloroprene has a (-Cl) group whereas natural rubber has a (-CH3) group. Polychloroprene rubber has good fire resistance and chemical resistance. So, these rubbers are more suitable for fire extinguishers.
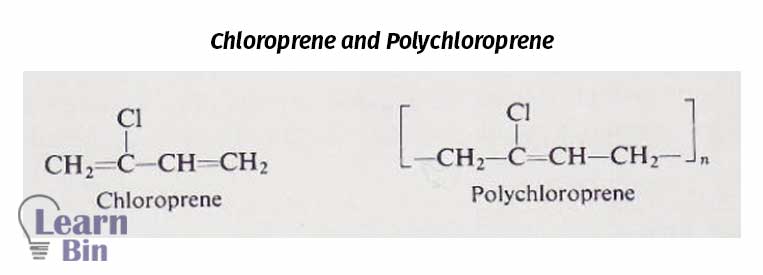
Poly-chloroprene rubber can be cross-linked to give strong vulcanizates. But it cannot be crosslinked by Sulphur as natural rubber. So, vulcanization is usually achieved by heating with metal oxides (CaO, MgO).
Like butadiene individual chloroprene units can be incorporated into polymer chains in various ways giving a polymer with different isomeric structures. The extent of the structural regularity of the poly-chloroprene chain affects the extent and rate of crystallization of the polymer.
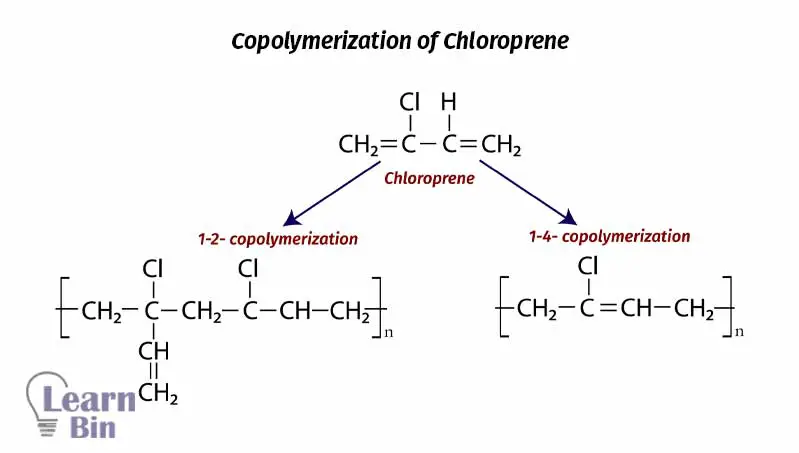
The extent of crystallization affects the physical properties of the polymer. As it increases, the polar properties like cohesive strength, modulus, tensile strength, hardness, resistance to permanent set, and resistance to swelling in hydrocarbon solvents increase sharply but elongation at break decreases. Due to the bulky groups of 1-2 copolymerized polychloroprene, they tend to have amorphous properties. 1-4 copolymerized polychloroprene has more crystalline properties.
A wide range of polymer properties can be obtained by varying the degree of stereo-regularity of the polymer, the molecular weight distribution of the polymer, the crosslink concentration in the polymer gel, or the gel content of the polymer. When a polymer is dissolved in a solvent and if it does not dissolve, the undissolved crosslinked particles are called gel content.
Functionalized forms of synthetic rubbers of both rubbery and glassy polymers are produced in the industries. The most important type of such rubbers is the Carboxylated synthetic lattices. Carboxylation is done by incorporating miner amounts of carboxylic acid groups into the polymer chain.
Eg; Carboxylated Styrene-Butadiene Rubber lattices.

mcpolymers.com - What Is the Difference Between Carboxylated and Non-Carboxylated?
The cover image was designed using a photo by Mark.murphy, available in Wiki | This work has been released into the public domain by its author, Mark.murphy at the Wikipedia project.