More results...


Fresh latex undergoes various processing steps before making products. Only natural rubber latex is undergone latex preservation and concentration steps. Just after the latex is collected from the tree, preservatives are added. Without adding preservatives, latex cannot be kept in liquid form; it will be coagulated. Preservatives are added to avoid coagulation. In the natural rubber latex industry, latex preservation is a must.
Field latex is preserved using a suitable preservative agent for long-term storage. Latex preservation is the process which maintaining the electrostatic stability of the latex within a period. Two changes occur shortly after latex leaves the tree. Which are “Spontaneous coagulation” and “Putrefaction”. A field latex sample coagulates after a few hours if it is un-preserved. Two theories have been proposed to explain the spontaneous coagulation of natural rubber latex.
Due to the development of acidity of latex through the interaction of various microorganisms with various non-rubbers in the latex. These acids release H+ ions to the latex.
So, the minus charge around the rubber particle is neutralized. Due to the destabilization of electrostatic stability, rubber particles are coagulated.
Due to the liberation of fatty acid anions through the hydrolysis of various lipid substances present in the latex and those fatty acid anions are then adsorbed onto the particle surface. Then they interact with the divalent metal ions such as Ca2+, Mg2+ which are present in latex initially. Divalent ions make interactions between rubber particles. This makes the rubber particles to be coagulated.
The acceptability of these two theories can be assessed by considering some experimental observations.
The pH of the fresh NR latex should be reduced below about 5 for coagulation by acidification. But the pH of the latex does not fall below 6.3-6.0 during spontaneous coagulation. Furthermore, spontaneous coagulation is not prevented by maintaining the pH at about 7 by the addition of a dilute alkali (base).
The process of spontaneous coagulation can be accelerated by the addition of a small amount of carboxylic soap. However, larger additions cause retardation in the process. Because the higher addition of carboxylic acid increases the minus charge density. So, the divalent ions are not enough to neutralize the minus charge. Therefore, the remaining minus is enough for the Brownian motion. The range of soap addition over which the coagulation time is a minimum depends on the Ca2+ and Mg2+ ion content of the latex.
Spontaneous coagulation depends on the presence of divalent ions such as Ca2+, Mg 2+. If these metal ions are absent, then the unpreserved latex can be kept in fluid condition for a longer period until putrefaction starts. These observations suggest the second theory is more acceptable.
An ideal preservative should have certain properties, it should
Ammonia preservation is widely used in the latex industry as a preservative. Ammonia preservation is two types of high ammonia method and low ammonia method. In the high ammonia method, the latex is added only 1% of Ammonia (NH3) by volume of latex. The low ammonia preservation system is also known as the LATZ method. In the LATZ method latex is added 0.25 – 0.25% of two other ingredients which are TMTD (0.0125%) and ZnO (0.0125%) by volume of the latex.
Ammonia plays several roles in latex. It acts as an antibacterial, as alkali, and as a metal ion deactivator. By reducing the bacteria content of the latex, ammonia helps to prevent the generation of acidity in the latex. And, it has basic properties, it helps to increase the pH level of the latex. Ammonia forms an ion complex with Mg2+ and deactivates its activity which leads to the coagulation process.
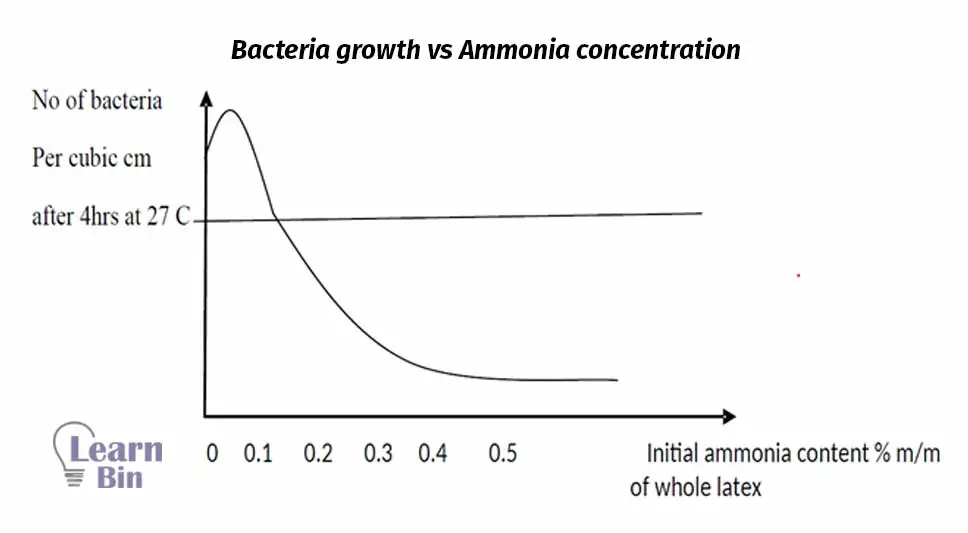
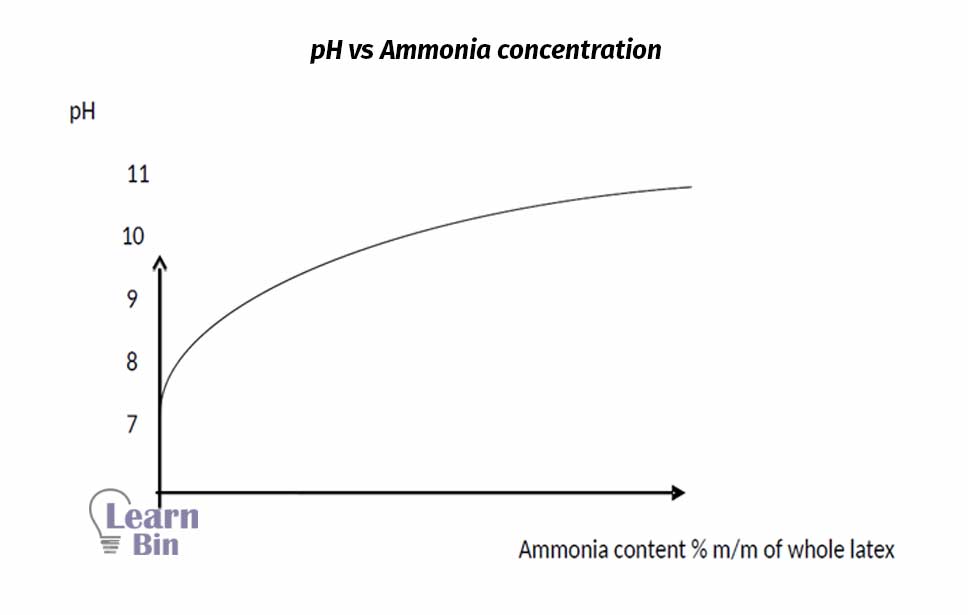
The major disadvantage of ammonia is that it interferes with some established latex processes. Ammonia should be handled either as cylinders of the anhydrous liquid or as concentrated aqueous solutions. Neither is very convenient. Because of handling difficulties, NH3 is not very cheap.
Additionally, ammonia gives a strong odor to the latex.
01. Formaldehyde and ammonia
Formaldehyde can preserve latex for a short period by killing bacteria. But for long-term preservation, ammonia should be added after.
02. KOH
Adding KOH increases the pH level of the latex.

Celkau.in - Latex preservation and concentration
The cover image was designed using a photo by M.arunprasad, available in Wiki | This file is licensed under the Creative Commons Attribution-Share Alike 3.0 Unported license.
Hello mis. Saumya Madubhashini. I am thankful for your article. I have few more question about field latex preservation.. Please help.
Please whatsapp me - []
Or mail - [] .thank you
Thank you for your feedback. If you have further question please send them to [email protected]