More results...


Solid materials can be divided into metals and non-metals. Theories on the structure and electronic properties of solid materials have been developed since early in the 20th century. In 1900 German physicist Paul Drude introduced a model that provides a quick practical way to form a simple picture and a rough estimate of the properties of a metal.
Drude has developed his theory of electrical-thermal conductivity based on the kinetic theory of gases. There are a few assumptions used in the kinetic theory of gas.
But in metal, there are two types of particles. One is positively charged (protons) and the other one is negatively charged (electrons). So, Drude assumed that protons are immobile and are attached to the heavier mass. Therefore, only electrons are free to move. Further, he assumed valance electrons are de-attached from the atoms and freely move through the metal. By applying the kinetic theory of gases, he assumed that the electrons freely move through solid material like gas molecules.
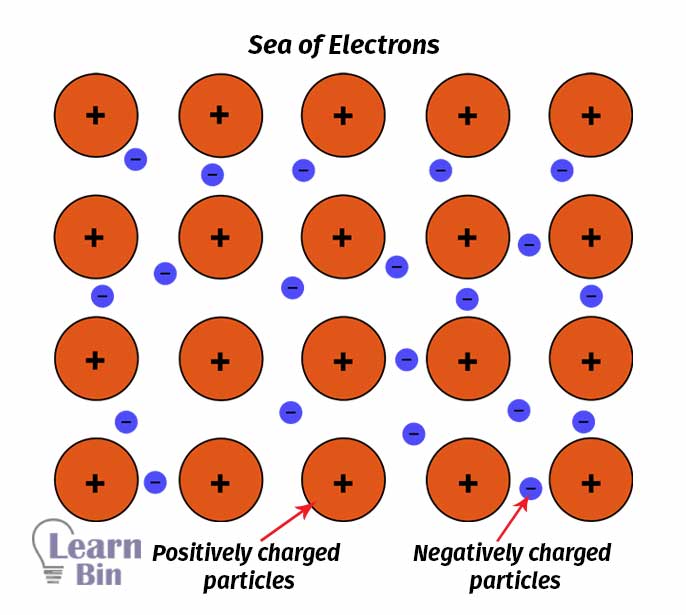
When two electrons collide, the interaction between other ions (electron and proton) is negligible. Therefore, in absence of an electric field, the electrons will move randomly in straight lines. The collisions are instantaneous. And it will alter the velocity of the electron.
Drude's theory explains the transport properties of electrons in metals. According to the Drude model, the number of electrons per cubic centimeter (the number density) can be estimated.
The number of elements per mole is NA = 6.022 × 1023 (Avogadro number). Let the atomic mass of the element “A”. If the mass density of the element is “ρm”, the number of moles per cubic centimeter is given by,
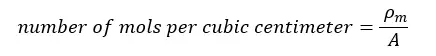
If the atom contributes “z” number of electrons, the number of electrons per cubic centimeter or number density of electrons (n) is given by,
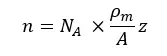
If we assume that the atoms are spherical, the radius of an electron (r) is given by,
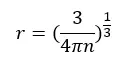
According to the Drude model, in the absence of an electric field, the charge carriers (electrons) are moving freely through the material. While they are moving throughout the material, they are just like gas molecules. When the electrons move around, they collide with the stationary atoms within the material. The average distance they traveled before colliding is called the “Mean free path (λ)”.
In the presence of an electric field (E), electrons start to move in the opposite direction of the electric field. While electrons are traveling under an electric field, they still collide with stationary atoms. The average time between two collisions is called “The relaxation time (τ)” as the electrons move, they will be moving with a certain velocity and this velocity is called “The drift velocity (Vd)”
According to “Ohm’s law”
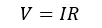
Where V is the voltage, I is the current and the R is the resistance. As well, the following equations can be obtained, where A is the cross-sectional area, and l is the length of the conductor.
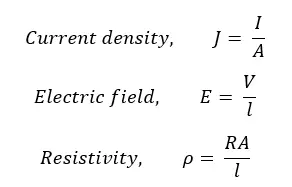
Using the above three equations, the Electric field can be expressed as,
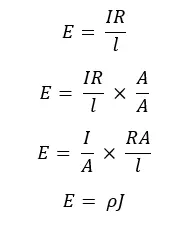
In terms of the electric conductivity (σ), the resistivity is given as,
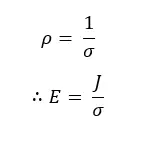
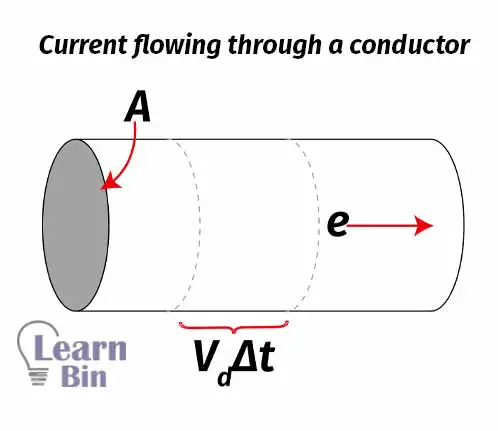
When charged particles flow under an electric field, an electric current is obtained. The electric current is the quantity of charged particles that travels in a unit volume per unit of time. It can be expressed as follows.

Where Vd is the drift velocity.
According to the Drude model, the net force acting on the electrons is given by,
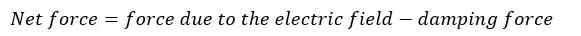
The damping force is the force that causes electrons not to move and make them collide with atoms. This force is occurred due to the positively charged particles in the metal. According to Newton’s third law,
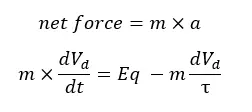
Where,
In a steady state, the acceleration is going to be zero. Therefore, the equation simplifies as follows.
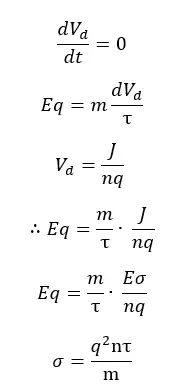
According to the above equation, the electrical conductivity is proportional to the square of charge carriers, the number of charge carriers, and the relaxation time. And it is inversely proportional to the mass.

The cover image was created using Image by Analogicus from Pixabay
Figure 01: Designed using an image by Muskid, licensed under CC BY-SA 3.0, via Wikimedia Commons