More results...


In a few of our previous articles, we have discussed the thermal expansion of solids. (Linear expansion of solids, area expansion of solids, and volume expansion of solids). In this article, we will discuss how liquids expansion happens when they are subjected to a change in temperature.
Thermal expansion of liquids is the increase in volume of a liquid when the temperature is increasing.
The rate of vibrations of molecules in a liquid increases with the temperature. Then the collisions between molecules increase. Therefore, molecules in the liquid push each other, and the intermolecular space is increasing.
Thus, the total volume of the liquid is increasing. Liquids show much more expansion than solids because the intermolecular interaction of liquids is lower than solids.
The thermal expansion of the liquids cannot be measured directly. Liquids cannot hold itself as a solid. Therefore, liquids should be contained in a container. There are two types of thermal expansion of liquids.
When heating, both the liquid, and the container are expanding. We can only observe this expansion which is a combination of the liquid and the solid container.
This expansion is known as the apparent volume expansion which is calculated without considering the expansion of the container.
Mathematically, if a liquid with the volume of V1 increases its volume up to V2 when the temperature is increased by Δθ,
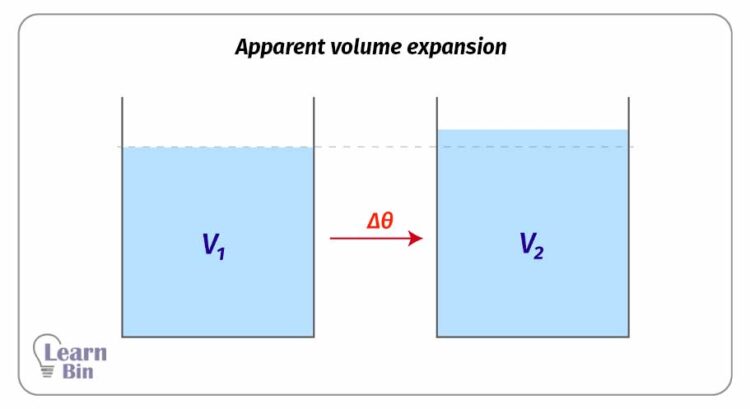
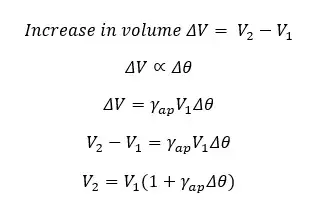
Where,
The apparent volume expansion coefficient is the apparent rational increase in the volume of the liquid when the temperature is increased by 1 °C or 1K.
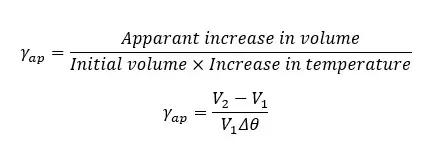
Absolute volume expansion is calculated by considering the expansion of the container.
Mathematically, if a liquid with the volume of V1 increases its volume up to V´ when the temperature is increased by Δθ,
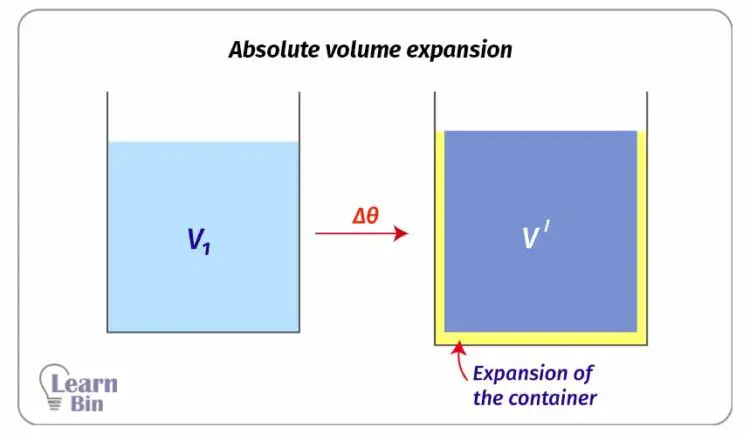
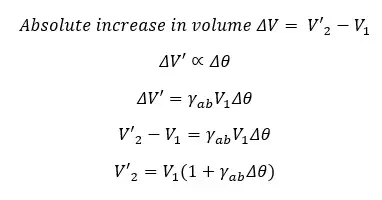
Where,
The absolute volume expansion coefficient is the absolute rational increase in the volume of the liquid when the temperature is increased by 1 °C or 1K.
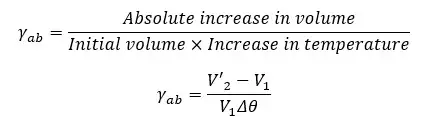
Absolute volume expansion is the sum of both the expansion of the container and the apparent volume expansion of the liquid. Absolute volume expansion is always higher than the apparent volume expansion.

Due to the thermal expansion of liquids, the volume of the liquid increases with the temperature. Therefore, the density is decreasing when the temperature is increasing.
Mathematically, if a liquid with the volume of V1, the mass of m, and the density of ρ1, increases its volume up to V2, when the temperature is increased by Δθ, the density of the liquid is written as follows.
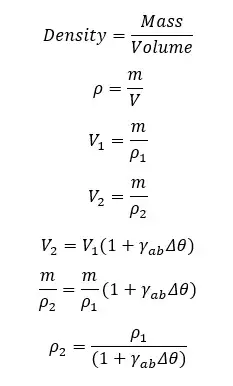
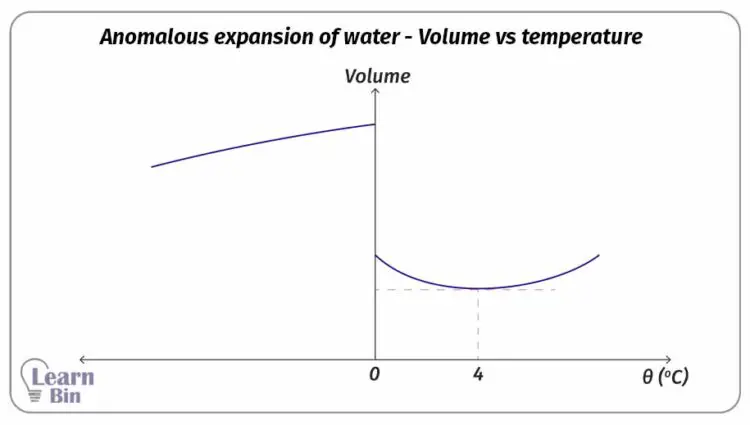
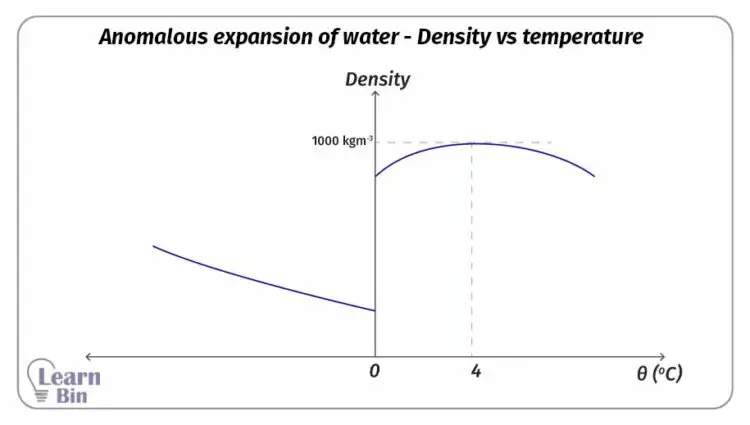
Due to the thermal expansion of liquids, their volume is increased when the temperature is increasing. But when the temperature of water is increased from 0 °C to 4 °C, water is contracting instead of expanding. (The volume is decreasing).
At 4 °C, water has the smallest volume and the highest density that can be obtained. At 4 °C the density of pure water is 1000 kgm-3.
The figure 02, and Figure 03 show the change in volume and the density of water when the temperature is increased from -10 °C to +30 °C.

The cover image was created using an image from PxHere licensed under CC0 Public Domain