More results...

Emulsion polymerization is a special case of free radical polymerization in a heterogeneous system.
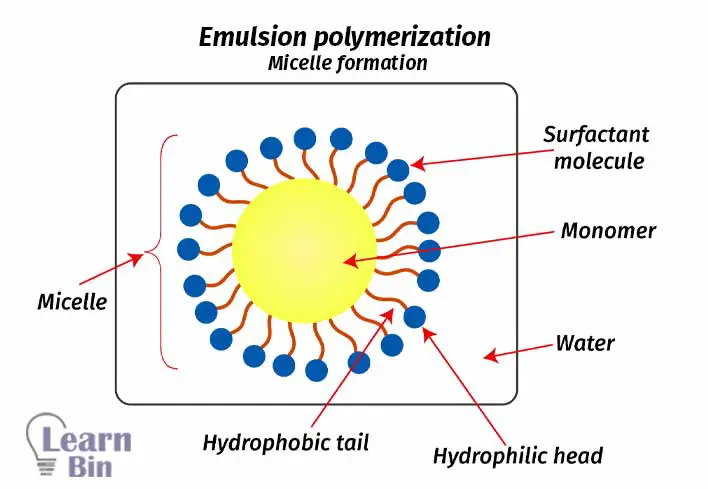
A water-insoluble monomer is dispersed and stabilized in an aqueous medium using surfactants. Monomer droplets that are surrounded by surfactant molecules are called micelles. Inside micelles, polymerization occurs.
There are four types of polymerization techniques bulk polymerization, solution polymerization, suspension polymerization, and emulsion polymerization. In the earlier stages, the bulk polymerization technique has been used widely. The major disadvantage of bulk polymerization is the lack of heat transfer efficiency in the reaction media. it may cause exploration and degradation of polymers. And there is a wide molecular weight distribution in polymer molecules.
To overcome the issues with bulk polymerization, the solution polymerization technique has been developed. In solution, polymerization, monomers, and initiators are dissolved in an inert solvent and let the polymerization occurs. The major disadvantage of solution polymerization is that results in polymers with low molecular weight.
Suspension polymerization is a heterogeneous polymerization technique where water-insoluble monomers are dispersed in an aqueous medium by mechanical agitation. In other words, it is like a bulk polymerization system that has been broken down into numerous droplets. Inside droplets, polymerization occurs. But the disadvantage of suspension polymerization is it needs continuous agitation. Because the droplets are not stable.
To overcome the disadvantages of each polymerization technique, the emulsion polymerization technique has been developed. The polymerization occurs at stabilized monomer droplets which are called micelles. Because the micelles are stable, it does not need continuous agitation as in suspension polymerization. Since there is a solvent (water) in the medium, the heat transfer efficiency is high. And emulsion polymerization yields high molecular-weight polymers.
The emulsion polymerization system is a heterogeneous system with two phases. Water-insoluble monomers are dispersed in water and stabilized using surfactants. Surfactants are molecules that have both hydrophobic and hydrophilic parts in them.
When preparation of the emulsion, monomer droplets are surrounded by surfactant molecules. The hydrophobic tails (hydrocarbon chain) of surfactants are arranged in the direction of monomer droplets and the hydrophilic heads are arranged in the direction of water. the monomer droplets that are surrounded by surfactant are called a micelle.
There are two methods as direct method and the indirect method that prepare an emulsion.
There are several mills are used in the industry to prepare emulsions. Such as colloid mills, ball mills, pebble mills, etc.
Emulsion polymerization is merely a bulk polymerization but in a finely divided state. In true emulsion polymerization, we can see the following special features.
In the initial steps of the polymerization monomers can be found monomers in three locations. Which are
Most monomers can be found in micelles (around 105 per milliliter). Except in highly soluble monomers, monomers can be hardly found in emulsified droplets and dissolved in water. Mostly the polymerization occurs in the micelles. These micelles are isolated from each other by electrostatic repulsion. In the second stage of polymerization (when the polymerization starts) monomers can be found in,
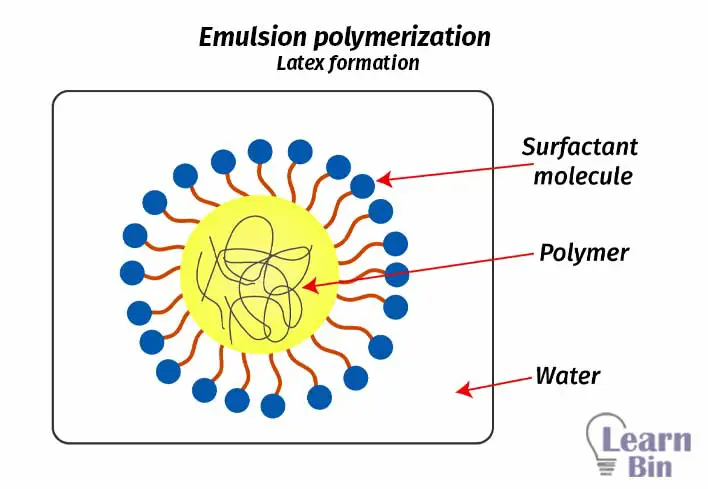
The polymerization starts once an initiator enters the micelles. The propagation of the chain continues until another initiator enters the same micelle. So, the chain length can be controlled by changing the rate of entry of radicals to the micelle. If the rate of entry of initiators is low, the chain length is high. As well growing polymer chains are located in different particles. So, those chains cannot terminate each other.
This results in a higher concentration of growing chains. Therefore, the rate of polymerization in emulsion polymerization is high. So, emulsion polymerization has a higher polymerization rate, and it yields polymers with higher molecular weight than homogeneous polymerization systems (bulk and solution polymerization).
| Homogeneous polymerization | Emulsion polymerization | |
| Monomer concentration (M) | 5 | 5 |
| Radical concentration (M) | 10-8 | The molecular weight of the polymer |
| Rate of polymerization per hour at 60℃ | 2 | 100 |
| The molecular weight of the polymer | 105 | 107 |
* The concentration of the monomer of the emulsion polymerization is the concentration within a micelle.
In an emulsion polymerization system, the amount of surfactant molecules is the main factor that controls the size of micelles. If there are high amounts of surfactant the size of micelles is low. Also, the number of micelles is high. Chain size mainly depends on the time between the entry of radicals into the micelles. This time depends on the number of latex particles (micelles).
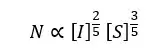
Where [I] is the concentration of initiators and [S] is the concentration of surfactants.

The cover image was created using an image by zeeshan ahmad from Pixabay
Dear madam,
We are manufacturing insulating rubber gloves and recently found the gloves having high failures/ punctures during the dielectric testing. I also noticed the tensile strength drop from 28mpa to 25mpa. However if i dry further in the oven with additional temp the tensile increase slightly but not as high as before. What are the possible reasons and any advise for me to improve the strength and dielectric strength.
btw, i am using natural rubber latex (PV).