More results...


Polyamides are derived from the poly-condensation of Amino-acid. Amino acids are connected by amide bonds. Polyamides are found as both natural and synthetic. Proteins are naturally occurring polymers. The first synthetic Polyamide was introduced commercially in 1938. It was Nylon. Nylon was developed in order to replace silk.
One of the earliest uses of nylon 6,6 fiber was for ladies' stockings and toothbrush bristles. During World War II, nylon was used as a war material such as parachutes, tire cords for bombers, ropes, flak jackets, and jungle clothing.
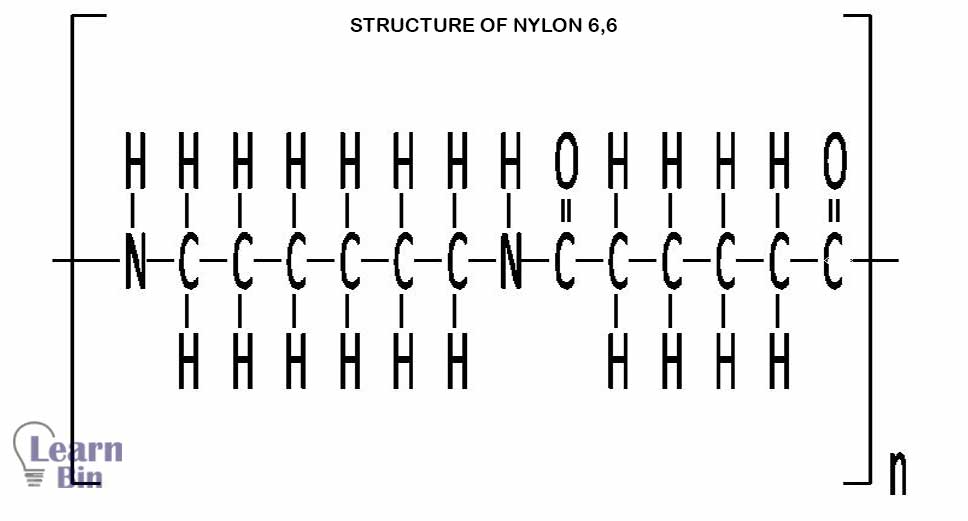
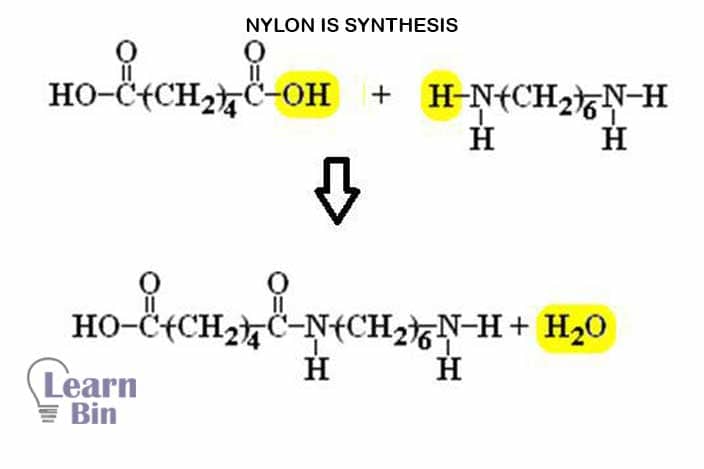
Nylon is synthesized by polycondensation of di-acid and di-amine.
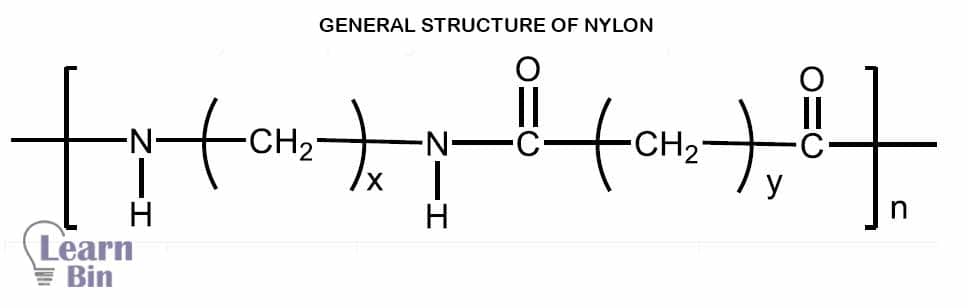
A variety of nylon grades are available commercially. Those are Nylon 6, Nylon 66, Nylon 6/6-6, Nylon 6/9, Nylon 6/10, Nylon 6/12, Nylon 11, and Nylon 12. The nomenclature of nylon is depending on the number of carbon atoms in di-acid and di-amine.
| Carbon atoms in di-amine | Carbon atoms in di-acid | Name |
| 4 | 6 | Nylon 4,8 |
| 4 | 7 | Nylon 4,9 |
| 4 | 9 | Nylon 4,11 |
| 6 | 4 | Nylon 6,6 |
| 6 | 6 | Nylon 6,8 |
| 6 | 9 | Nylon 6,11 |
Polyamides are thermoplastic materials. Most of the polyamides tend to be semi-crystalline and it shows lamella structure. Polyamides are linear chains with a low molecular weight of around 15000 g/mol.
The tensile strength of polyamides is upon to polar groups and crystalline structure of the polymer. Polyamides have a high density of hydrogen bonds. These hydrogen bonds are formed between the oxygen atoms of the carbonyl group and hydrogen atoms in polyamide. The high amount of hydrogen bonding influences the stiffness, crystallinity, flexibility, glass transition point, and gas permeability of the polyamide. The tensile strength of nylon is 600 kg/cm2.
Polyamides are the most common and important commercial examples, of thermoplastic materials that are capable of competing with metals. When considering polyamides, polyamide is stronger than steel.
The melting temperature of polyamide is 255℃ -265 ℃. The high melting temperature has advantages and disadvantages. The advantage is Polyamides can be used in high-temperature environments. Heat-stabilized systems allow sustained performance at temperatures up to 185 ℃ (for reinforced systems). A disadvantage of a high melting point is, that it should be supplied with high energy to be melted at the processing stage.
Polyamides tend to provide good resistance to Hydrocarbons. Polyamides swell in alcohols and dissolve in phenols. Polyamides can be attacked by strong acids, alcohols, and alkalis. Polyamides are good dielectric in an oily medium.
Polyamides have good resistance to abrasion.
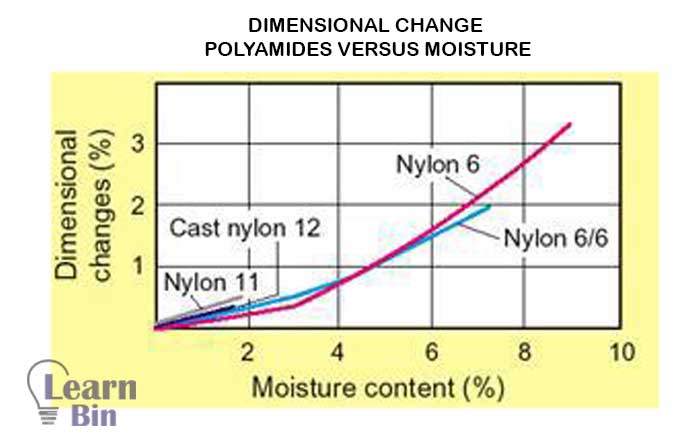
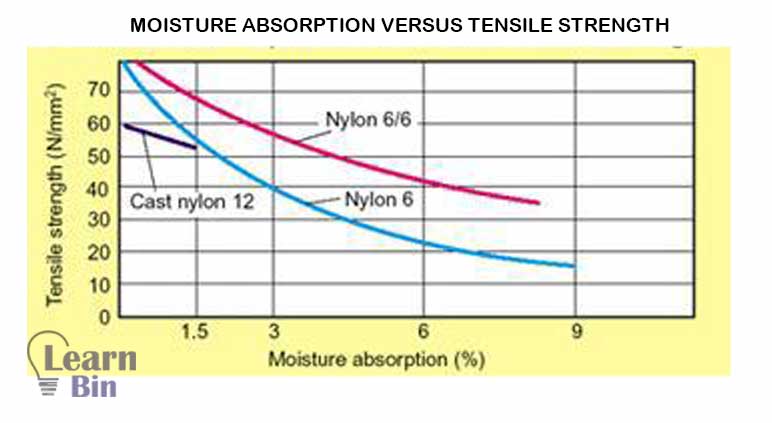
A disadvantage of polyamide is it tends to absorb moisture from its surroundings. This absorption process will happen until equilibrium is attained and that can have a negative effect on "dimensional stability". And it also affects the tensile strength.
Aramids are a class of synthetic polyamides that are formed from aromatic monomers and yield fibers of exceptional strength and thermal stability. The tensile strength of aramids is about 2.5 times of nylon fibers.
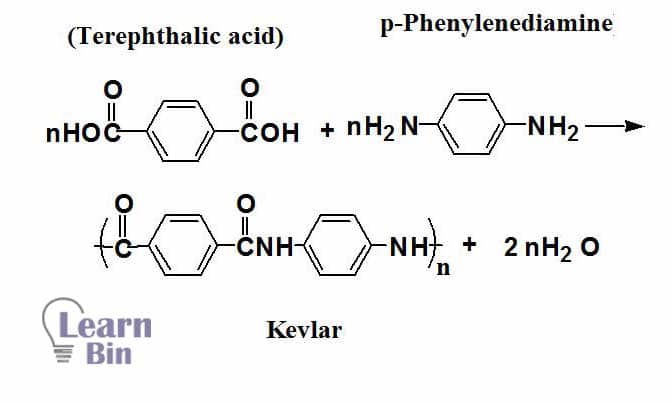
One of the well-known examples of aramids is Kevlar. Kevlar is a polyaromatic amide (an aramid). Kevlar is synthesized by polymerization of Terephthalic acid and p-phenylenediamine.
Cables of Kevlar are as strong as cables of steel, but only about 20% of the weight. Kevlar fabric is used in the production of bulletproof vests, jackets, and raincoats.
Raw polyamide is blended with different types of additives such as Heat and light stabilizers, Plasticizers, Lubricants, Fillers, and Pigments in order to increase its properties.
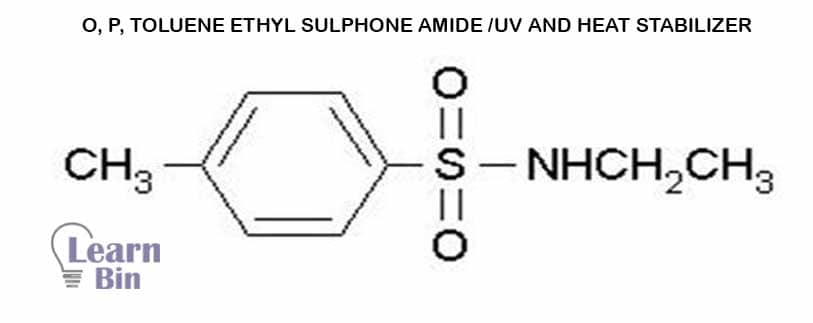
Heat and light stabilizers are used to protect PA against oxidation and UV for outdoor applications.
Fillers can be blended with raw polymer up to 60%. Fillers are added to the material for two main reasons. The first one is to reduce the cost. Low-cost fillers are blended with raw materials. in order to increase the volume. This type of filler is called “Non-reinforcing filler”. Graphite, Chine clay, and Talc are some non-reinforcing fillers. They decrease the cost of compounds and improve water resistance.
The other reason is to increase the tensile strength of the material. These filters are called “Reinforcing fillers”. Asbestos and glass fibers are reinforcing fillers. They increase the strength of the material. Glass fibers increase the tensile strength of the material by about -3000kg/cm2
Glass fiber-reinforced nylon is a better alternative to steel. Short glass fibers (2mm-5mm length) or Long glass fibers (10-12mm length) are used as reinforcing material.
Nylon can be processed into final products by conventional plastic processing methods. Such as injection molding, extrusion, blow molding, rotational molding, and thermoforming.
Nylon is extruded at 220-300℃ temperature. Nylon films and fibers are manufactured by extrusion. The main drawback of nylon extrusion is its moisture content. Due to the polar groups of nylon, it tends to absorb moisture. A too-high content of moisture results in bubbles/foaming of the melt and its results in holes in the film. The maximum allowable moisture content is 0.1 % for the extrusion of nylon.
Nylon fibers are used in textiles, fishing lines,s and carpets. Nylon film does not transmit smell and is resistant to hydrocarbons. Therefore, Nylon films are used for food packaging, offering toughness and low gas permeability. Nylon shows high resistance to temperature. Therefore, nylon films are used for boil-in-the-bag food packaging. As well Nylon films are also used for the sterilization of hospital equipment.
Electrical insulation, corrosion resistance, and toughness make nylon a good choice for high-load parts in electrical applications such as insulators, switch housings, and ubiquitous cable ties. Another major application is for power tool housings.


The water content of about 0.15% to 0.2% is preferred in nylon pellets used in injection molding.
To ensure proper water content levels in the injection molding of nylon it is necessary that the granules be dry. Therefore, the polymer should be dried in an oven at about 70-90°C. The drying time should be 10 to 48 hours.
Nylon melts at 255℃ -265 ℃. High temperature accelerates oxidation.
Molding and extrusion compounds find many applications as replacements for metal parts, for instance in car engine components. Nylon has self-lubricating properties that make it useful for gears and bearings.


Cover Image was designed using PublicDomainPictures from Pixabay