More results...


In an equilibrium system where a chemical reaction is occurring, if there is no net change in the concentrations of reactants and products, such a condition is called chemical equilibrium.
The equilibrium constant gives an idea about the relationship between the products and reactants in the media at a given temperature. Let’s take the following chemical equilibrium system,
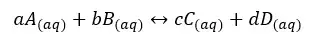
In the above reaction, A, B, C, and D are the reactants and products. a, b, c, and d are the stoichiometric ratios of each component. For the above reactions, the equilibrium constant (Kc) for concentration can be written as follows.
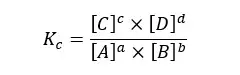
Where,
For the above system equilibrium constant can be expressed according to the mole fraction of each component.
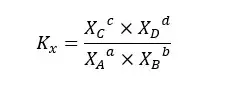
Where,
If the system is a gaseous system, the equilibrium constant for pressure (Kp) can be written as follows,
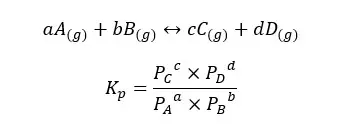
Where,
Kc, Kx, and Kp are only dependent on the temperature.
01. Haber process reaction
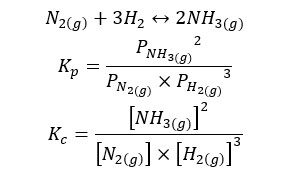
02. Partial dissociation of Sulfur trioxide
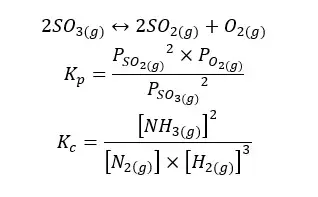
03. Esterification reaction equilibrium
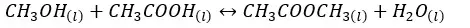
The above system is a liquid equilibrium system. So, in this system, Kp is not considered.
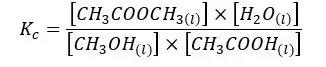
If the above system is in an aqueous medium, and water is formed from the reaction, the concentration of the water remains constant.
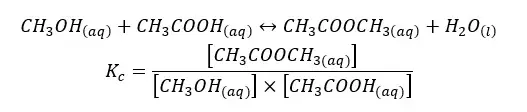
04. Ammonium hydrogen sulfide dissociation
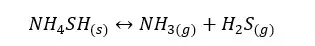
In the above system, NH4SH is solid. Therefore, its concentration remains constant. So, the concentration of NH4SH is not included in the Kc expression. And a solid does not affect the pressure of the system.
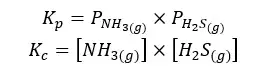
05. Calcium carbonate dissociation
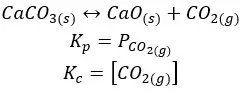
06. The reaction between iron and water
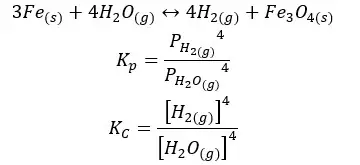
07. Dissociation of Phosphorous pentachloride
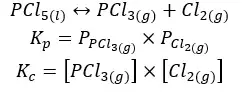
In the above system, PCl5 liquid has separated from the gaseous phase. Therefore, the concentration of PCl5 is constant.
Let’s take the following reaction,
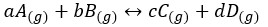
For the above system, Kc and Kp can be written as follows,
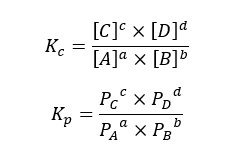
If all the above gases are ideal, the concentration of each component can be expressed as follows. To derive these expressions, the ideal gas law is used.
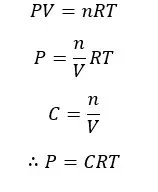
Where,
According to the above equation, the partial pressure of each component can be written as follows,
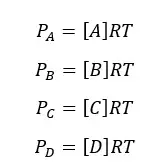
Where [A], [B], [C], and [D] are concentrations of each component, and PA, PB, PC, and PD are partial pressure of each component. Let’s substitute the above expressions to the Kp equation,
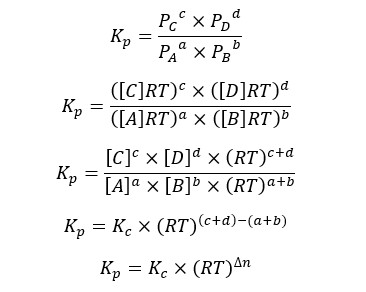
Where Δn is the difference between the sum of the stoichiometric ratios of products and the sum of the stoichiometric ratios of reactants.
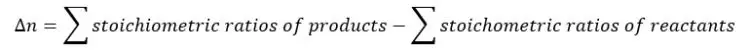
If one of the products or reactants is a liquid or a solid, the stoichiometric ratio of that component is not considered.

The cover image was created using an image by RF._.studio from Pexels